
Compliance Assurance
Stay compliant with industry regulations and standards.
Cost-effective
Achieve regulatory success with Elexes, all within your budget.

Elexes provides end-to-end CE Marking consulting services to help you meet EU MDR and IVDR requirements seamlessly. Whether you manufacture medical devices, IVDs, or combination products, our CE Marking consultants assist you with regulatory strategy, technical documentation, conformity assessments, and notified body interactions.
Ready to obtain CE certification without stress?
Let’s make your device EU-ready.
CE marking is your product's passport to the European Economic Area (EEA). It's not just a logo, it's a declaration of safety, compliance, and quality. Here’s what it means for your business:
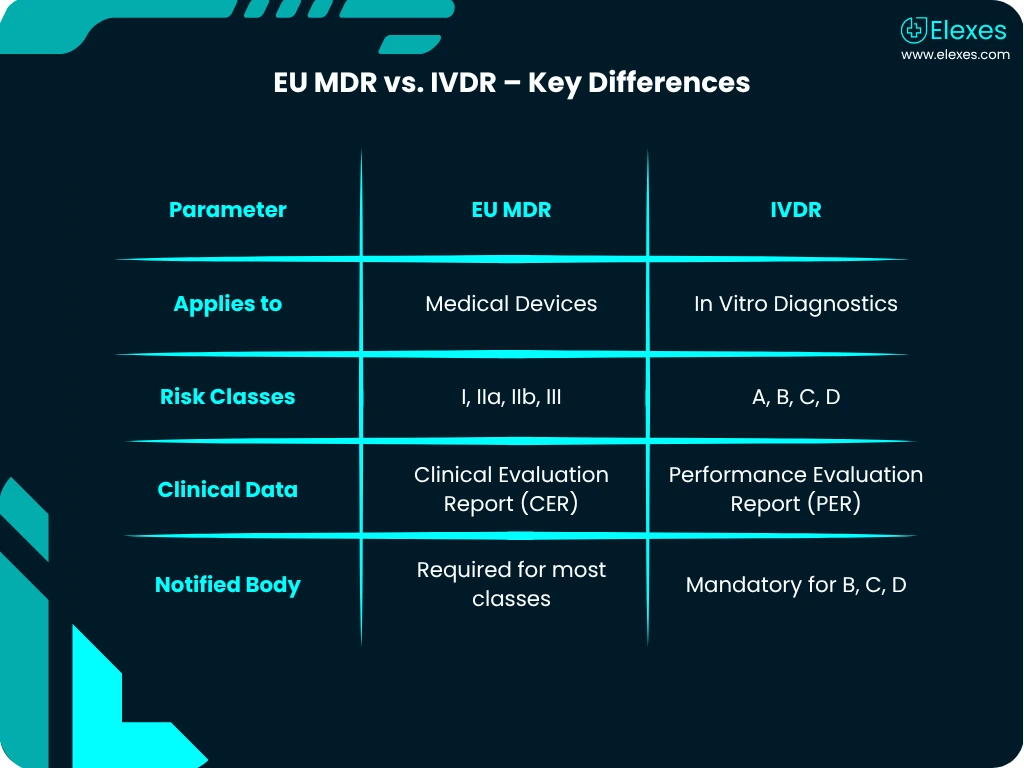
All medical and in-vitro diagnostic devices sold in the EU must comply with EU MDR or IVDR. CE marking confirms this compliance.
Products with CE marks are seen as safe, reliable, and high-quality, leading to faster adoption in hospitals and clinics.
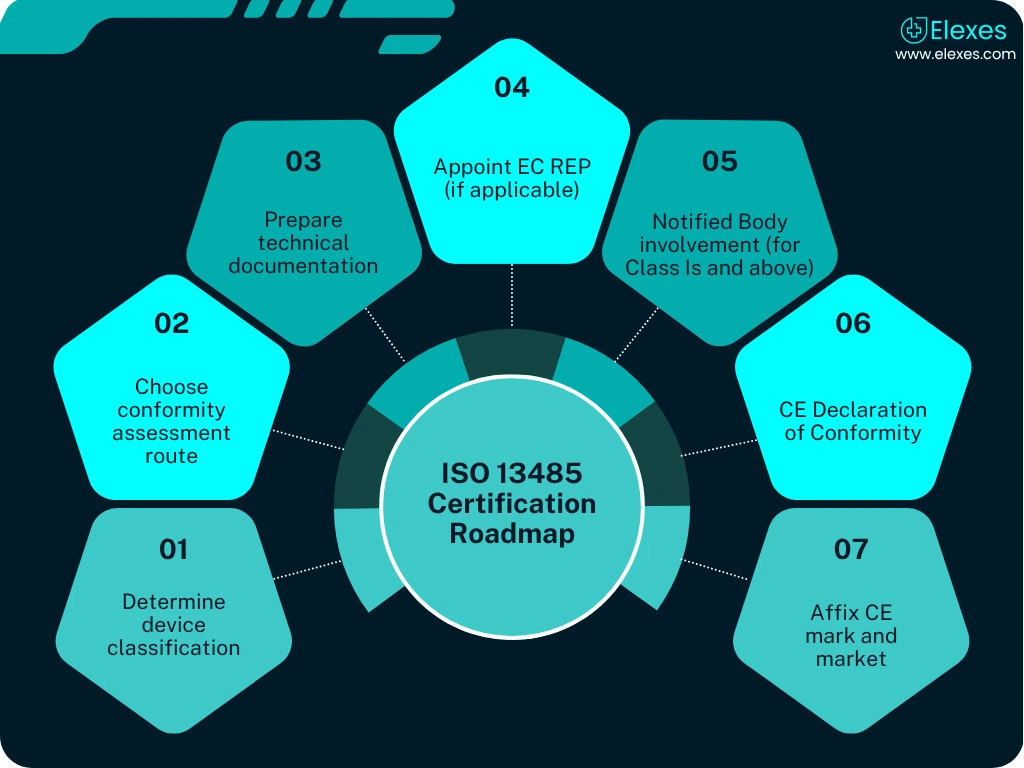
From GSPR mapping and risk analysis to notified body reviews, CE marking requires precise documentation and strategic execution.

⦿ Regulatory Strategy Development: Device classification, roadmap, and compliance path.
⦿ Technical File Creation: GSPR checklist, risk management (ISO 14971), usability, clinical data.
⦿ IVDR or MDR Gap Assessments: Evaluate existing files and remediate gaps.
⦿ Notified Body Liaison: Submission support, Q&A responses, and audit prep.
⦿ Labeling & UDI Compliance: Review and develop EU-compliant labels and instructions.
⦿ Authorized Representative (EC REP) support: Representation for non-EU manufacturers.
Also explore our EU MDR Consultant and IVD Regulatory Consulting services.
⦿ Regulatory pathway analysis
⦿ Clinical evaluation planning
⦿ Classification confirmation
⦿ Technical file preparation services
⦿ Design dossier review
⦿ Annex-specific documentation (Annex II, III)
⦿ PMS and PSUR planning
⦿ Vigilance reporting systems
⦿ CAPA planning & implementation

We’ve helped startups and multinational companies across the globe secure CE marks for diverse products, from Class I reusable surgical tools to Class III implants and cutting-edge IVD platforms.
💡 With our CE certification consultants, you don’t just check boxes, you enter the market confidently.
Let’s Get Your Device CE Certified!
It’s not just about getting a CE mark; it’s about staying compliant, competitive, and confident. Partner with Elexes to take the complexity out of compliance.
CE marking for medical devices is a certification that indicates a device meets the health, safety, and environmental protection standards required in the European Economic Area.
Yes, especially for Class II and III devices or IVDs. A CE Marking consultant helps navigate EU MDR/IVDR requirements, technical files, and notified body submissions.
It can take 6–12 months depending on device classification, documentation readiness, and notified body timelines.
Stay compliant with industry regulations and standards.
Achieve regulatory success with Elexes, all within your budget.
Experience timely results with our efficient services.
We offer 100% confidentiality understanding how critical the data is for you.
We offer different services that will help you not only keep your product well in boundaries of regulations but also speed up the entire approval process. Some of these services are -








CEO Masterlink, Arizona

CEO Novasignal, Los Angeles

President ViDava, Florida

Sr. Exe Treedental, Hong Kong

Manager Outset Medical, California

CTO Jana Care, Massachusetts

MD Blackrock Pharma, England

VP Regulatory AliveCor, California

Owner Liz Inc., Arizona

CEO Radformation, New York


























Never miss out on any important update on the regulatory & compliance industry across the globe. Subscribe to our newsletter now.
Copyright 2025, Elexes Medical Consulting Pvt. Ltd. All Rights Reserved
Working Hours : Monday to Friday 9:00 AM - 7:00 PM