
Compliance Assurance
Stay compliant with industry regulations and standards.
Cost-effective
Achieve regulatory success with Elexes, all within your budget.
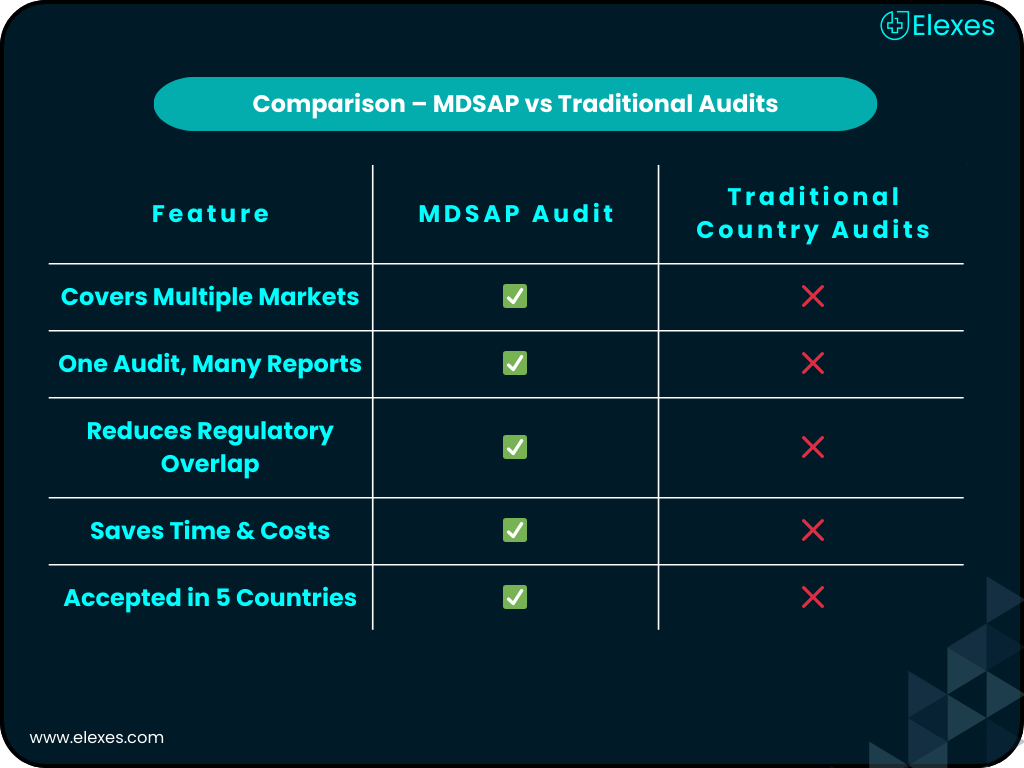
The Medical Device Single Audit Program (MDSAP) allows manufacturers to meet multiple regulatory requirements through a single audit. Elexes simplifies your MDSAP certification journey by helping you implement and maintain a compliant, inspection-ready Quality Management System (QMS). Whether you're aiming for access to the U.S., Canada, Australia, Brazil, or Japan, we’ll make your path smooth and strategic.

MDSAP streamlines audits and boosts your credibility in key global markets. With regulators like the FDA, Health Canada, and TGA on board, it’s more than a certification, it's a passport to international compliance. For manufacturers expanding globally, MDSAP certification is no longer optional; it’s essential.
At Elexes, we offer:
⦿ Tailored QMS implementation aligned with ISO 13485 and MDSAP
⦿ Gap assessments and internal audits
⦿ Audit readiness and regulator-specific consulting

We begin with an in-depth gap analysis, comparing your current QMS against MDSAP audit model requirements. From there, we tailor a roadmap that closes compliance gaps and prepares your team for the audit. We provide documentation support, training, and ongoing guidance, all personalized for your product type and target markets.
Elexes has helped Class I, II, and III device manufacturers across diagnostics, SaMD, electromechanical devices, and implantables meet MDSAP standards confidently.
MDSAP is recognized by five key regulatory authorities. Here’s what it means for your business:

Reduces the need for duplicative inspections and shortens timelines for ANVISA registration.
We evaluate your existing QMS and highlight medical device regulatory compliance gaps with the MDSAP audit model, using ISO 13485:2016 as the core.
Our experts draft or update your procedures, work instructions, and forms aligned to regulatory expectations of each participating authority.
We conduct internal audits and simulate real MDSAP inspections to ensure you’re fully prepared and confident.
We provide hands-on training for your quality and regulatory teams to handle MDSAP auditor interviews and document requests.
From planning to audit closure, we assist with documentation, CAPA responses, and direct interactions with auditing organizations.
Stay compliant after certification with periodic updates, QMS maintenance, and assistance during surveillance audits.
Let Elexes guide you through every phase with clarity and precision. Below are two resources to help you understand what it takes to become MDSAP compliant.

Let Elexes help you succeed with MDSAP Certification.
MDSAP Certification is a quality system audit recognized by regulators in the US, Canada, Brazil, Australia, and Japan, allowing one audit to meet multiple requirements.
Yes, since January 2019, Health Canada mandates MDSAP certification for all medical device manufacturers selling in Canada.
The full MDSAP process can take 4–8 months, depending on the state of your QMS and audit readiness.
Stay compliant with industry regulations and standards.
Achieve regulatory success with Elexes, all within your budget.
Experience timely results with our efficient services.
We offer 100% confidentiality understanding how critical the data is for you.








CEO Masterlink, Arizona

CEO Novasignal, Los Angeles

President ViDava, Florida

Sr. Exe Treedental, Hong Kong

Manager Outset Medical, California

CTO Jana Care, Massachusetts

MD Blackrock Pharma, England

VP Regulatory AliveCor, California

Owner Liz Inc., Arizona

CEO Radformation, New York


























Never miss out on any important update on the regulatory & compliance industry across the globe. Subscribe to our newsletter now.
Copyright 2025, Elexes Medical Consulting Pvt. Ltd. All Rights Reserved
Working Hours : Monday to Friday 9:00 AM - 7:00 PM