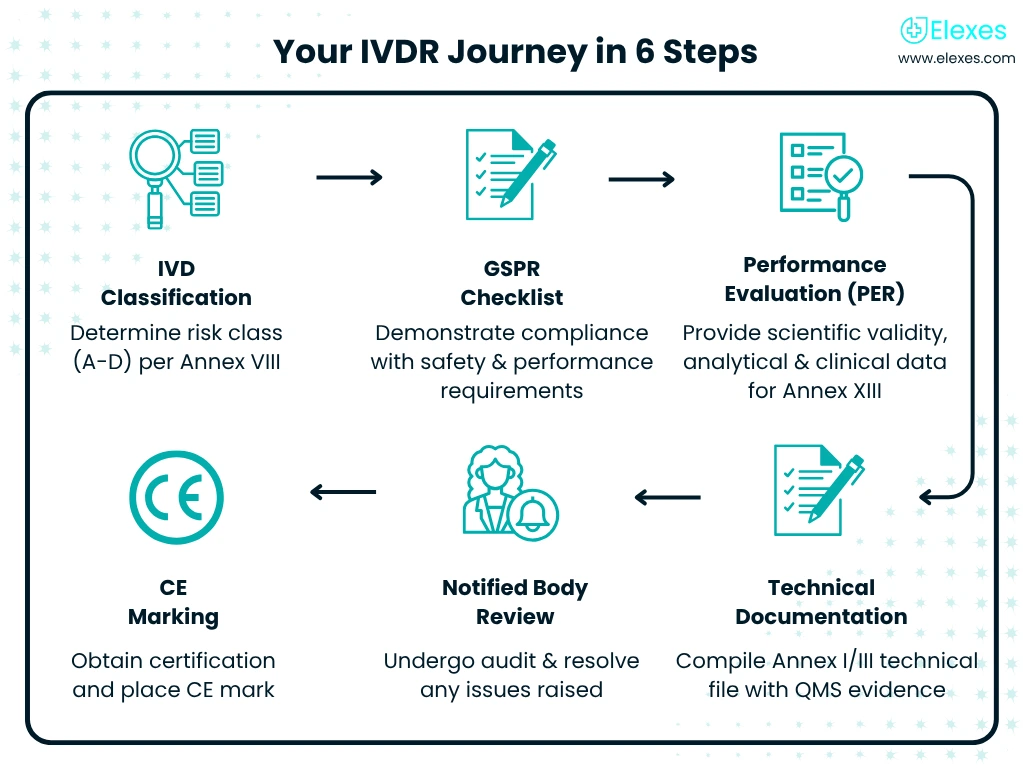
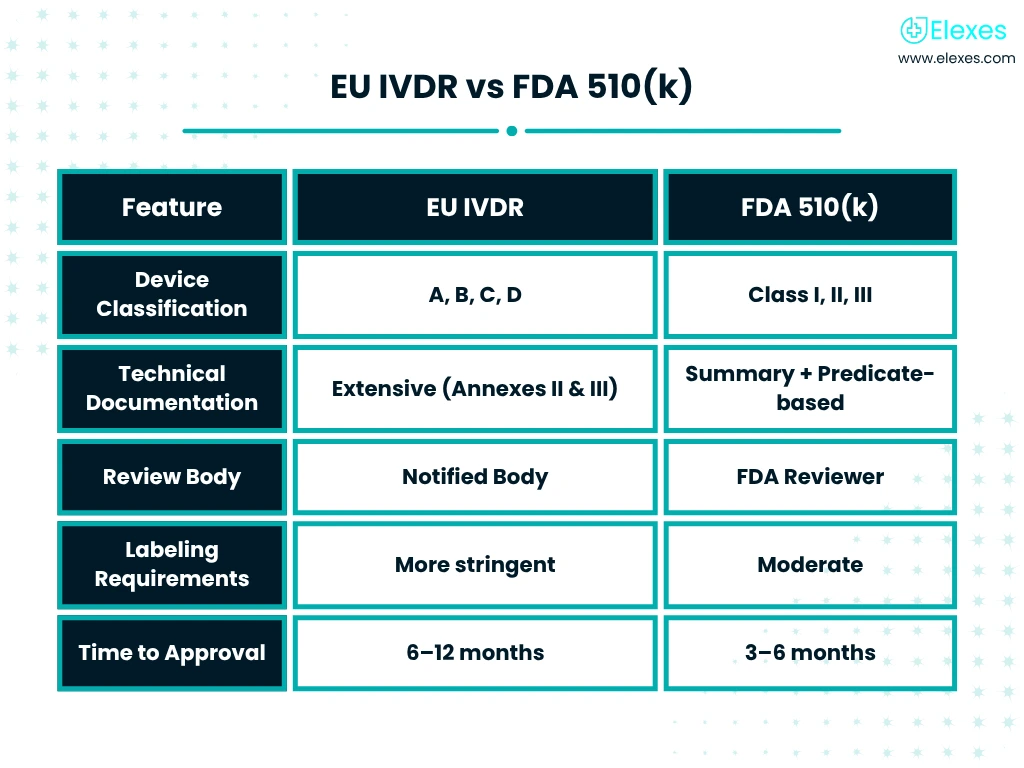
A Trusted Team of Consultants
Elexes offers a globally recognized ISO 13485 certification consultant who is renowned to enhancing your business credibility in the medical devices industry.
Local Language
Auditor
Our presence across the globe ensures audits are conducted by local-language auditors, facilitating clear communication and understanding.