
Compliance Assurance
Stay compliant with industry regulations and standards.
Cost-effective
Achieve regulatory success with Elexes, all within your budget.

Gaining FDA clearance through the 510(k) pathway is essential for most Class II medical devices in the U.S. market. At Elexes, we guide you through every step, from determining substantial equivalence to preparing the submission and interacting with the FDA. Our experienced team of FDA 510(k) consultants ensures that your device complies with all regulatory expectations and reaches the market faster.

With a proven track record of successful 510(k) clearances across multiple device categories, including IVDs, SaMD, wearables, and implantable devices, Elexes brings in-depth regulatory expertise and a client-centric approach. We align your submission with the latest FDA guidance, identify the right predicate, and ensure your documentation is audit-ready.
We work with startups, SMBs, and Fortune 500 MedTech firms to:
⦿ Eliminate guesswork in device classification and pathway selection
⦿ Identify the optimal predicate device
⦿ Prepare a compliant, streamlined 510(k) dossier
⦿ Manage FDA Q-Sub and RTA interactions
⦿ Accelerate time to clearance

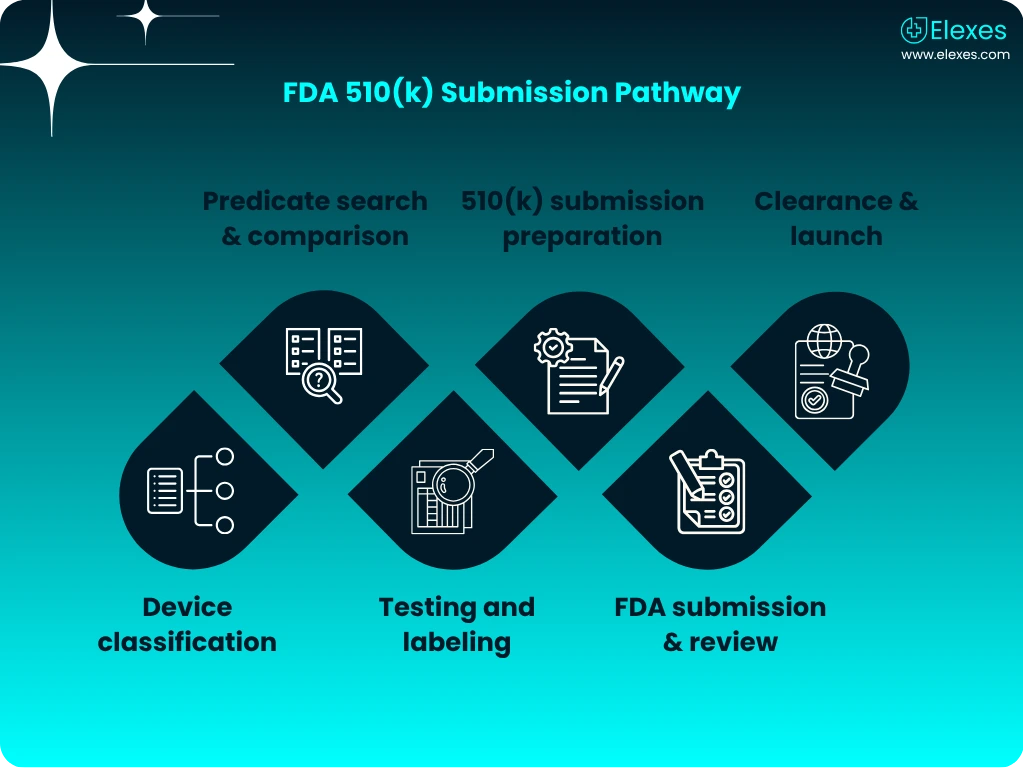
The 510(k) process is designed to demonstrate that your device is “substantially equivalent” to a legally marketed device. Elexes walks you through every phase:
⦿ Regulatory Strategy: Clear direction on classification and pathway
⦿ Predicate Analysis: Robust scientific and technological comparisons
⦿ Documentation Preparation: Technical file, labeling, test reports, and more
⦿ Submission: Format per RTA Checklist & eSTAR or traditional 510(k)
⦿ FDA Interaction: Responses to AI letters, deficiencies, and additional info
⦿ Clearance: Post-clearance support for labeling and commercialization
Our team provides comprehensive support, from discovery to clearance:

Elexes ensures that your submission isn’t just complete, it’s compliant, strategic, and submission-ready. Let Elexes be your partner in successful 510(k) clearance.
Explore how 510(k) compares with other FDA pathways and get a clear view of the step-by-step 510(k) submission journey.
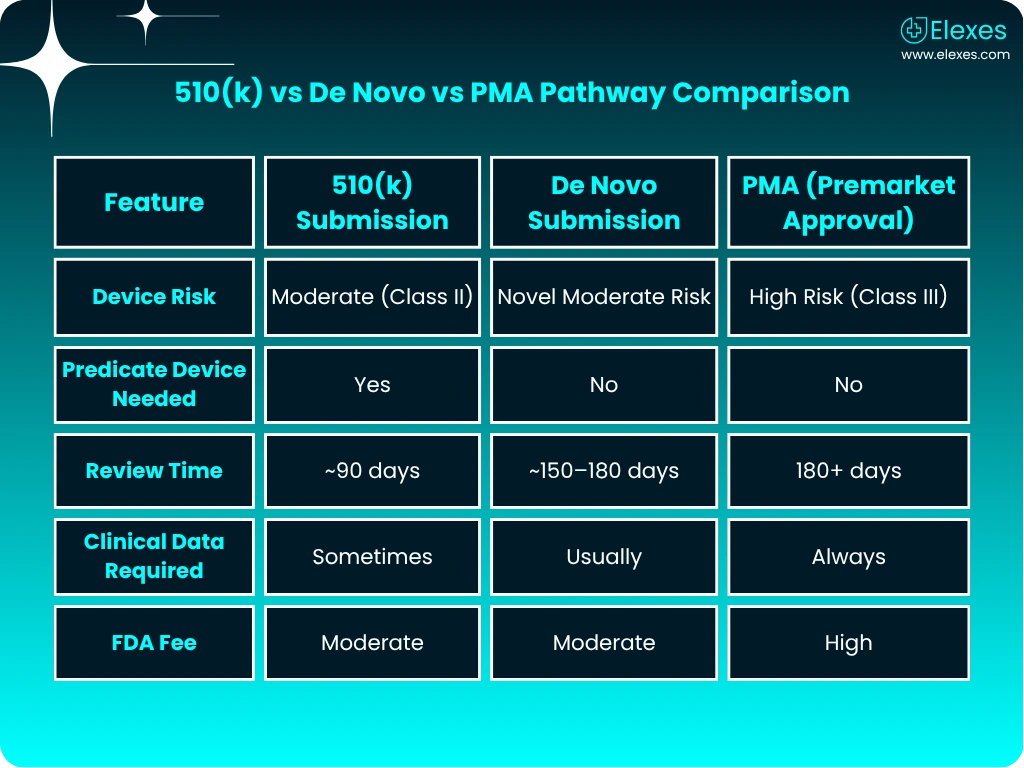
A 510(k) submission is a premarket notification to the FDA to demonstrate that a medical device is substantially equivalent to a legally marketed predicate device.
The FDA typically reviews 510(k) submissions within 90 calendar days, but the process may take longer based on device complexity or additional information requests.
Manufacturers of most Class II and some Class I devices must submit a 510(k) before marketing their product in the U.S.
Delays often result from incomplete documentation, poor predicate choice, or failure to meet testing standards. Elexes helps avoid these pitfalls.
Stay compliant with industry regulations and standards.
Achieve regulatory success with Elexes, all within your budget.
Experience timely results with our efficient services.
We offer 100% confidentiality understanding how critical the data is for you.
We offer different services that will help you not only keep your product well in boundaries of regulations but also speed up the entire approval process. Some of these services are -








CEO Masterlink, Arizona

CEO Novasignal, Los Angeles

President ViDava, Florida

Sr. Exe Treedental, Hong Kong

Manager Outset Medical, California

CTO Jana Care, Massachusetts

MD Blackrock Pharma, England

VP Regulatory AliveCor, California

Owner Liz Inc., Arizona

CEO Radformation, New York


























Never miss out on any important update on the regulatory & compliance industry across the globe. Subscribe to our newsletter now.
Copyright 2025, Elexes Medical Consulting Pvt. Ltd. All Rights Reserved
Working Hours : Monday to Friday 9:00 AM - 7:00 PM