Predicate Device Selection for 510(k): FDA Strategy & Common Pitfalls
Introduction
Bringing a medical device to market through the FDA’s 510(k) pathway requires more than just technical know-how—it starts with selecting the right predicate device. A predicate serves as the benchmark against which your subject/new device is evaluated for safety, effectiveness. But even with the summary about the devices and hundreds of such devices listed in the FDA’s database, identifying the most suitable predicate can be a daunting task. In this blog, we’ll walk through what a predicate device is, why it’s so critical, and how to select a predicate device confidently to strengthen your 510(k) submission.
Elexes has helped 100+ medical devices gain 510(k) clearance with strategic predicate device selection and strong SE justification. If you’re preparing a 510(k), choosing the right predicate can make or break your clearance strategy. Here’s what you must consider.
What is a Predicate Devices?
A predicate device is a one that is already marketed in the US and that can be used to compare with a new device that the manufacturer plans to launch in the US market. According to the FDA, a predicate device can be:
⦿ A device legally marketed prior to May 28, 1976 (pre-amendments device).
⦿ A device reclassified from Class III to Class II or I.
⦿ A device that has been found substantially equivalent through the 510(k) process.
Nevertheless, an important point is to emphasize that not identical devices would be acceptable as long as differences do not raise new questions regarding safety and effectiveness of the product.
FDA Criteria for Predicate Device Selection
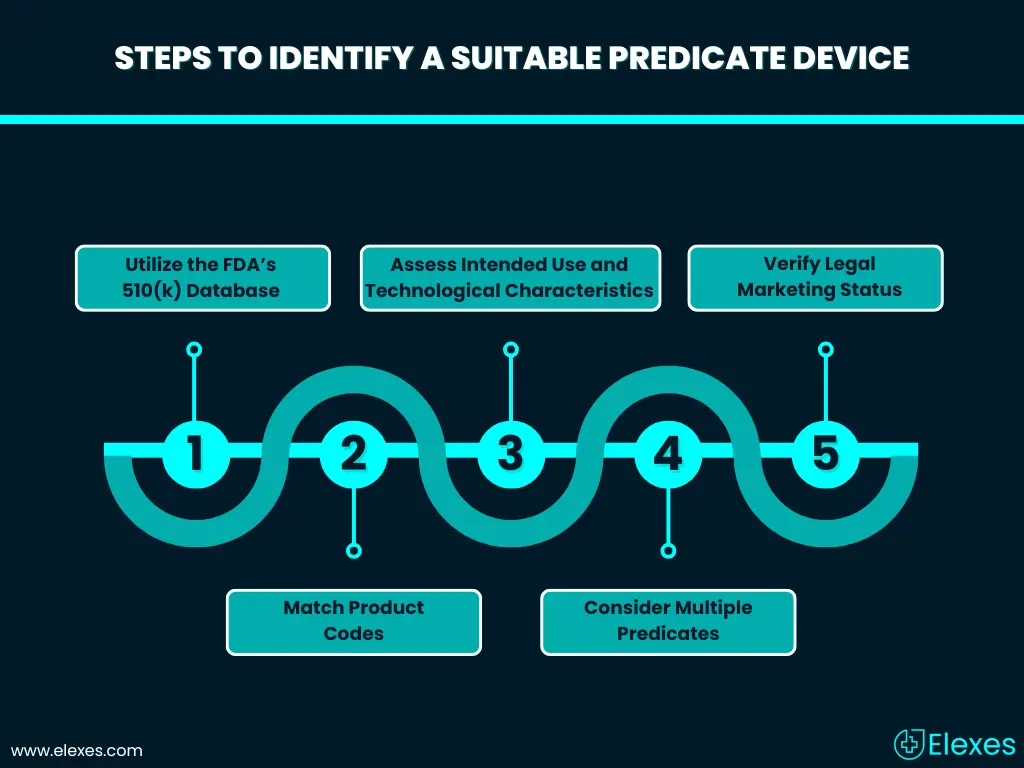
1. Utilize the FDA’s 510(k) Database
Potential predicate devices can be identified using the FDA’s 510(k) database. This database contains information of the devices which have been cleared through 510(k) process. Individuals can search the predicate device in the database by using different parameters such as device name, product code or the manufacturer. The 510(k) predicate device database is revised on a regular basis and it is important for manufacturers to stay updated with the latest information.
2. Match Product Codes
Use predefined product codes to narrow down the search to devices that are technically similar to the new product.
3. Assess Intended Use and Technological Characteristics
The primary predicate device should closely match the new device in terms of intended use, technology, and performance characteristics. Differences in technological characteristics are permissible only if they do not raise new questions of safety and effectiveness. A clear rationale must be provided to support these differences.
4. Consider Multiple Predicates
In some cases, a new device may draw comparisons to more than one predicate device. While the FDA requires a primary predicate, secondary predicates can support the equivalence argument, particularly when demonstrating similarities in different aspects of the device.
5. Verify Legal Marketing Status
It’s imperative to confirm that the predicate device is still legally marketed and has not been subject to recalls or safety concerns. Using a device that has been withdrawn from the market or has known issues can jeopardize the 510(k) submission and may result in rejection or additional review To verify the regulatory status and safety history of the predicate device, use the recall database, MAUDE Database.
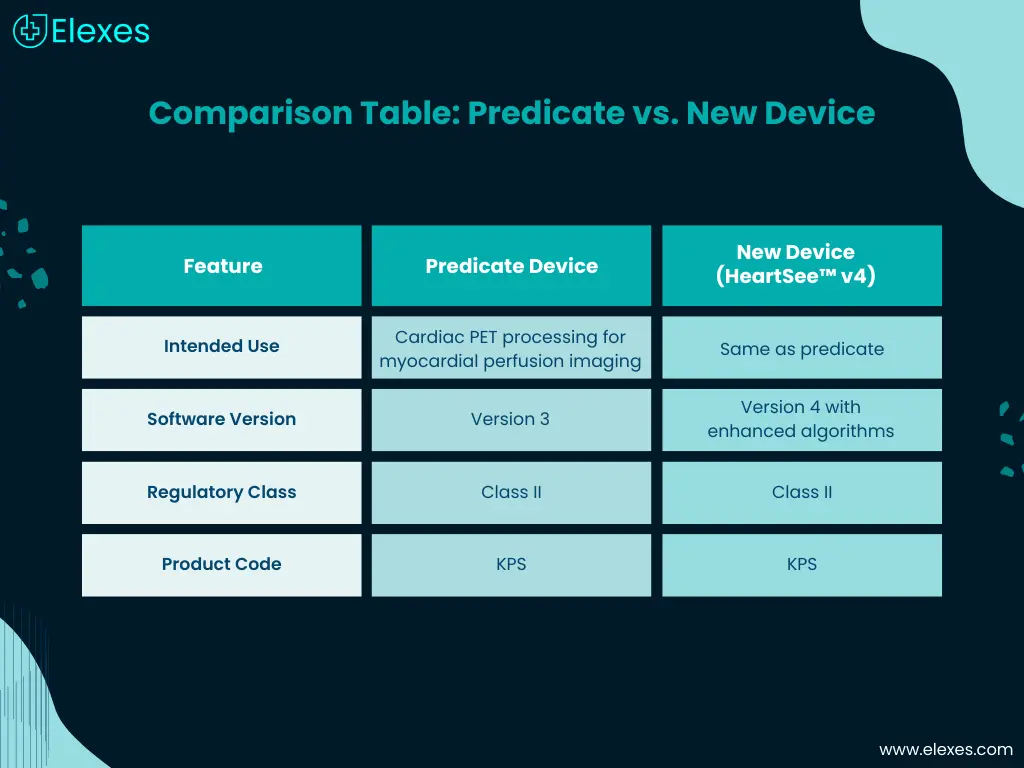
Comparison Table: Predicate vs. New Device
You can refer to a brief of 510(k) summary comparison table from one of the recent submissions here:
K243881 – 510(k) Summary
Common Mistakes in Predicate Selection
⦿ Relying solely on marketing claims: Always verify technical details through the FDA summary or labeling.
⦿ Using a device with incomplete public data: If performance specifications aren’t disclosed, it’s harder to argue substantial equivalence.
⦿ Selecting a very old device: Technological gaps may make it hard to justify equivalence, leading to extra testing.
⦿ Ignoring regulatory history: Use FDA Recalls Database to check if the predicate was involved in enforcement actions or recalls.
Expert Support: When and Why to Seek Help
While experienced regulatory professionals may navigate the FDA landscape comfortably, startups and new manufacturers may struggle to identify the best predicate. Engaging a regulatory partner like Elexes ensures:
⦿ Proper classification and product code selection
⦿ Accurate labeling of intended use
⦿ Comparative table creation with justification
⦿ Gap assessments and testing recommendations

Conclusion
Choosing the right predicate device is among the first steps towards the 510(k) submission. The more extensive the comparison and research into possible predicates, with emphasis, ensuring it lines up in purposes and technological characteristics and confirming that the intended change is legally marketed, the sounder the model of FDA substantial equivalence. Although engaging the regulatory experts will bolster the odds in favor of actual FDA clearance.
Need Help Choosing the Right Predicate?
FAQs
Can I use more than one predicate device?
Yes, FDA allows multiple predicates if each supports different aspects of substantial equivalence. A primary predicate must still be identified.
What if my device is novel or doesn’t match any predicate?
You may consider the De Novo pathway if no predicate exists and your device presents moderate risk. FDA will evaluate novelty and safety independently.
How important is labeling similarity in predicate selection?
Labeling must align in terms of indications for use, intended users, and environments. Major labeling differences can trigger a rejection for SE.
Is software considered part of technological characteristics?
Yes. FDA evaluates software algorithms, architecture, and performance in determining substantial equivalence between your device and the predicate.
Where can I find predicate device summaries?
You can use the FDA 510(k) database to search by product code, device name, or company to access summary statements of previously cleared devices.





















