
Introduction
ISO 13485 certification has become a critical investment for medical device companies seeking global market access. The demand for certified Quality Management Systems (QMS) in the medical device space has intensified following regulatory updates—most notably, the FDA's QMSR alignment with ISO 13485:2016, which takes effect February 2, 2026. Without a compliant QMS, companies risk losing access to the EU, Canada, Australia, and the upcoming modernized FDA regulatory framework.
Most manufacturers underestimate the true cost of ISO 13485 certification. Budget overruns are common, typically driven by three factors: underestimating internal implementation time, hiring consultants without medical device QMS expertise, and overlooking ongoing surveillance audit fees after initial certification.
This guide covers everything you need to plan your certification budget:
- Realistic cost ranges by company size
- Key factors that drive prices up or down
- A full breakdown of cost components
- How to build a more accurate budget for your certification journey
TLDR
- Total ISO 13485 certification cost typically ranges from $15,000–$40,000 for small companies to $100,000+ for large or complex organizations
- Three primary cost drivers: certification body audit fees, consultant fees, and internal implementation costs (staff time, training, infrastructure)
- Companies with higher device risk classifications (Class III, implantables, SaMD), design controls, and multiple sites face significantly higher costs
- Choosing a consultant with specialized medical device expertise reduces total cost by minimizing re-audits and rework
How Much Does ISO 13485 Certification Cost?
ISO 13485 certification does not have a fixed price. Costs vary substantially based on organization size, device complexity, existing QMS maturity, and certification scope.
Three budgeting mistakes account for most cost overruns:
- Underbudgeting for internal implementation time — The certification body audit fee is only 15–30% of total cost; staff time redirected to QMS documentation, training, and process changes is the largest expense
- Choosing low-cost generic consultants — Consultants without medical device expertise create systems that fail audits or can't support regulatory submissions, leading to costly re-audits and remediation
- Ignoring ongoing surveillance fees — Annual surveillance audits and 3-year recertification cycles are recurring costs that need day-one budget planning
The ranges below reflect total investment — including implementation, consulting, and audit fees — grouped by organization size.
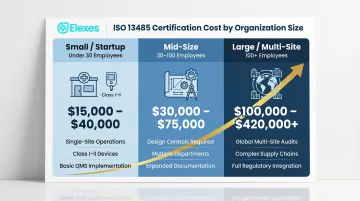
| Organization Size | Typical Total Cost | Key Scope |
|---|---|---|
| Small / Startup (under 30 employees) | $15,000–$40,000 | Single-site, Class I–II devices |
| Mid-Size (30–100 employees) | $30,000–$75,000 | Design controls, multi-product scope |
| Large / Multi-Site (100+ employees) | $100,000–$420,000+ | Multi-site, SaMD, multi-jurisdiction |
Small Company / Startup (Under 30 Employees)
Typical Total Cost Range: $15,000–$40,000
This tier covers Stage 1 and Stage 2 certification audit fees, basic QMS documentation support, limited internal auditor training, and single-site certification scope.
Best suited for:
- Startups and early-stage medical device companies
- Class I or II devices without design controls
- Distributors or IVD manufacturers with a simple, well-defined product scope
Mid-Size Company (30–100 Employees)
Typical Total Cost Range: $30,000–$75,000
Scope at this level expands significantly:
- Full QMS implementation support
- Design control documentation and risk management file development (ISO 14971)
- Internal auditor training and pre-audit gap assessment
- Certification audit fees across broader product scope
This range fits growing manufacturers with Class II–III devices, companies adding design and development activities, and organizations integrating ISO 13485 with FDA QMSR compliance.
Large / Complex Organization (100+ Employees or Multi-Site)
Typical Total Cost Range: $100,000–$420,000+
At this scale, costs reflect the full complexity of the certification program:
- Multi-site certification and full-scope QMS overhaul
- Extensive design controls with SaMD-specific requirements (IEC 62304 integration)
- Post-market surveillance systems
- Regulatory affairs alignment across FDA, EU MDR, Health Canada, and TGA
This tier applies to established manufacturers with high-risk devices, multiple product lines, and global distribution — particularly organizations pursuing MDSAP certification for simultaneous multi-jurisdictional access.
Where your organization falls within these ranges depends on factors beyond headcount alone. QMS maturity, device risk class, and certification scope each push costs up or down significantly — and those variables are worth mapping before engaging any certification body or consultant.
Key Factors That Drive ISO 13485 Certification Costs
Pricing is shaped by a combination of organizational, regulatory, and operational variables. Understanding each helps you avoid both overspending and costly underpreparation.
Device Risk Classification and Complexity
Higher-risk device classifications require more extensive documentation, design controls, and risk management files—directly increasing consultant hours and audit scope.
Risk classification shapes cost in three primary ways:
- Class III devices and implantables require comprehensive design validation, clinical evaluation data, and biocompatibility testing documentation—significantly expanding audit scope
- SaMD developers must integrate IEC 62304 software lifecycle requirements alongside ISO 13485, adding a distinct layer of documentation and cost
- Combination devices (drug/biologic/diagnostic) require cross-functional QMS processes spanning multiple regulatory frameworks simultaneously
Existing QMS Maturity
Companies starting from scratch face the highest implementation costs, while organizations with an existing ISO 9001 or partial QMS can significantly reduce documentation development time and consultant hours.
Organizations with ISO 9001 certification already have foundational QMS processes in place. Mid-size firms transitioning from ISO 9001 typically complete certification in 4–6 months versus 9–18 months for those starting from scratch. Existing document control, CAPA systems, and management review processes reduce consultant scope—and cost—substantially.
Certification Scope: Manufacturing vs. Design + Manufacturing
Organizations that only manufacture to customer specifications have a narrower scope than those performing full product design.
Design controls under ISO 13485:2016 Section 7.3 rank among the most resource-intensive requirements. Section 7.3 requires documented procedures across design planning, inputs, outputs, reviews, verification, validation, transfer, and change control—each with full traceability requirements.
Cost impact:
- Manufacturing-only scope may reduce total cost by 30-40%
- Design + manufacturing scope requires dedicated design control expertise
- Design History File (DHF) development represents 20-35% of implementation effort
Number of Sites and Geographic Spread
Each additional manufacturing, distribution, or development site increases audit days, travel costs, and documentation requirements.
IAF MD 1 sets the methodology for auditing multi-site organizations, including sampling approaches and audit time per selected site. Multi-site certifications can substantially increase both consultant and certification body fees—often adding $15,000–$40,000 per additional site depending on complexity.

Consultant Specialization and Engagement Model
Generic ISO consultants may charge less upfront but lack the medical device QMS expertise needed to pass first-time audits, leading to costly re-audits and remediation.
Specialized consultants bring two advantages generic ISO consultants cannot replicate:
- Deep familiarity with design controls, risk management under ISO 14971, and post-market surveillance requirements
- QMS architecture that supports FDA 510(k), CE marking technical files, and MDSAP audits—not just the certification itself
On engagement model: for smaller organizations, project-based consulting scoped to a defined certification path is typically more cost-efficient than retaining a full-time in-house quality manager.
Breaking Down the Total Cost of ISO 13485 Certification
The final certification invoice is only part of the picture. Total investment includes four distinct cost categories that companies must plan for both upfront and on an ongoing basis.
Certification Body Audit Fees
Initial certification audit fees are a one-time cost; surveillance and recertification audits recur on an annual and three-year cycle respectively.
What's included:
- Stage 1 audit (documentation review): $500–$3,000 for small companies; $2,000–$5,000 for mid-size
- Stage 2 audit (on-site implementation audit): $1,000–$8,000 for small; $8,000–$20,000 for mid-size
- Initial certification total: $10,000–$20,000 typical range
Ongoing audit fees:
| Company Size | Annual Surveillance | 3-Year Recertification |
|---|---|---|
| Small | $2,000–$5,000 | $4,000–$10,000 |
| Mid-Size | $5,000–$15,000 | $10,000–$25,000 |
| Large/Multi-Site | $15,000–$40,000 | $25,000–$60,000 |
Consulting and Implementation Support Fees
Consulting fees are primarily a one-time cost for initial certification, though ongoing QMS support may extend into annual retainer arrangements.
What's included:
- Gap analysis and readiness assessment
- QMS documentation development (procedures, work instructions, forms)
- Design control setup and DHF structure
- Risk management file creation (ISO 14971)
- Internal auditor training
- Mock audit and certification audit preparation
Cost ranges by company size:
- Small: $5,000–$30,000
- Mid-Size: $20,000–$60,000
- Large/Complex: $50,000–$100,000+
This is typically the largest and most variable cost component. Costs fluctuate based on your QMS maturity at the start, device complexity, and whether your consultant handles documentation build-out or only provides advisory oversight. Companies with more gaps at baseline and higher-risk device classifications will consistently land at the upper end of these ranges.
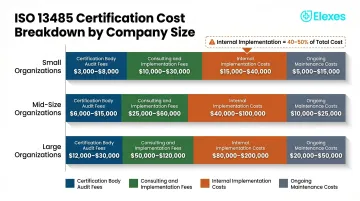
Internal Implementation Costs
Most internal implementation costs are one-time, though training refreshers recur as staff changes or procedures are updated.
What's included:
- Staff time (quality managers, engineers, operations personnel redirected to QMS work)
- Internal auditor training
- QMS software subscriptions ($10,000–$50,000 annually for eQMS platforms)
- Equipment calibration and validation
- Facility or process modifications
Cost ranges:
- Small: $8,000–$25,000
- Mid-Size: $25,000–$60,000
- Large: $60,000–$150,000+
These costs are frequently underestimated and can represent 40–50% of total investment.
A quality manager dedicating 50% of their time for 6 months to QMS implementation represents $30,000–$50,000 in internal labor cost alone.
Ongoing Maintenance Costs Post-Certification
One-Time / Recurring: Recurring (annual)
What's included:
- Annual surveillance audits
- Continual QMS improvement activities
- Periodic staff retraining
- Preparation for recertification every three years
- QMS software subscription renewals
- Document updates triggered by design changes or regulatory revisions
Estimated annual cost range: $15,000–$60,000 depending on company size and complexity
Low-Cost vs. High-Cost ISO 13485 Certification — What's the Real Difference?
The gap between budget and premium certification paths is not simply about spending more: it reflects real differences in audit readiness, consultant expertise, and QMS sustainability over time.
First-Time Audit Pass Rate
- Low-cost paths (generic consultants, minimal prep) carry higher risk of audit findings and re-audit fees
- Premium paths with medical device specialists and thorough gap analysis yield consistently higher first-pass rates
- Re-audit fees typically add $5,000–$15,000 and delay market access by 2–4 months
QMS Quality and Regulatory Utility
- Budget systems may pass certification but fail to support FDA submissions, EU MDR technical files, or MDSAP audits
- A well-built QMS serves as a regulatory asset across multiple markets — EU MDR Article 10(9) mandates a QMS proportionate to risk class, covering regulatory strategy, risk management, clinical evaluation, design, PMS, vigilance, and CAPA
- Premium implementations integrate with 510(k) submissions, CE marking technical documentation, and Health Canada licensing
Long-Term Maintenance Burden
- Cheaply built QMS documentation is harder to maintain, update during design changes, and defend in post-market surveillance reviews
- This creates higher ongoing costs — both direct (consultant remediation) and indirect (staff time lost to regulatory delays)
The "cheapest" certification path frequently becomes the most expensive once re-audit fees, regulatory submission failures, and QMS rebuilds enter the picture. Factoring in those downstream costs before choosing a path is where the real savings happen.
How to Estimate the Right Budget for ISO 13485 Certification
The right budget is not the lowest one—it is the one calibrated to your device risk class, target markets, organizational complexity, and long-term compliance goals.
Key factors to assess before budgeting:
Identify Target Markets Early
Each regulatory jurisdiction adds scope requirements that directly affect cost. Knowing your target markets upfront lets you build a single QMS that covers all requirements rather than retrofitting later.
- EU MDR, FDA QMSR, Health Canada (MDSAP), and TGA all have distinct but overlapping requirements
- Certifying for multiple jurisdictions simultaneously through a single QMS build is more cost-efficient than sequential certifications
Audit Your Current QMS Maturity Honestly
A pre-certification gap analysis — often available as a standalone engagement — gives you the clearest foundation for accurate cost estimation. Without it, mid-project surprises and scope creep are common.
- Avoids unplanned cost overruns from late-stage scope expansion
- Identifies high-risk areas (design controls, supplier management, CAPA) that need focused investment before the audit
Choose Consultants With Specialized Medical Device QMS Experience
Expertise spanning regulatory, quality, and clinical requirements minimizes re-audit risk. A consultant who has navigated FDA QMSR, EU MDR, and MDSAP across multiple product types will anticipate gaps that a general ISO consultant may miss.
Elexes has supported 250+ projects across medical devices and IVDs, with a 90% audit clearance rate — not just for initial certification, but through FDA submissions, EU MDR compliance, and MDSAP audits. That track record reflects the difference between a QMS built to pass one audit and one built to support the full regulatory lifecycle.
Common Budgeting Mistakes That Drive Up ISO 13485 Certification Costs
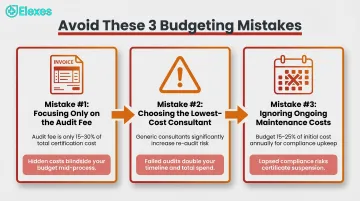
Most cost overruns don't come from surprises — they come from predictable mistakes made during planning. Here are the three that consistently inflate ISO 13485 certification budgets:
Focusing only on the certification body audit fee while ignoring the larger costs of consulting, internal staff time, and QMS infrastructure. The audit fee typically represents just 15–30% of total investment; implementation accounts for 60–75%.
Choosing a consultant based on the lowest quoted price rather than demonstrated medical device QMS expertise and audit clearance history. Generic ISO consultants unfamiliar with design controls, risk management under ISO 14971, or post-market surveillance requirements tend to build systems that fail audits.
Underestimating ongoing maintenance costs. Annual surveillance audits, staff turnover requiring retraining, and QMS updates triggered by design changes or regulatory revisions are recurring expenses. Plan for 15–25% of your initial certification cost every year.
Frequently Asked Questions
How much does ISO 13485 certification cost?
ISO 13485 certification typically costs $15,000–$40,000 for small companies (under 30 employees, simple devices) and $100,000+ for large organizations (multi-site, high-risk devices). Company size, device complexity, QMS maturity, and consulting scope are the main cost drivers.
How much does ISO 13485 certification cost for a small business?
Small businesses with simple device scope and fewer than 30 employees typically fall in the $15,000–$35,000 range. This usually includes Stage 1 and Stage 2 audit fees, basic QMS documentation support, and limited training—but often excludes internal staff time, which can add another $8,000–$25,000 in hidden costs.
How long does ISO 13485 certification take?
Most companies complete ISO 13485 certification in 6–12 months; complex or high-risk organizations starting from scratch may require 12–18 months. Key factors include existing QMS maturity (organizations transitioning from ISO 9001 can complete in 4–6 months), device risk classification, number of sites, and whether design controls are in scope.
Is ISO 13485 certification worth it?
Yes. ISO 13485 is a prerequisite for EU (CE Mark), Canadian, and Australian market access, and aligns with the FDA QMSR effective February 2, 2026. Most companies see ROI within 18–36 months through process efficiency gains and expanded market opportunities.
Can an individual be ISO 13485 certified?
No. ISO 13485 is an organizational certification for a company's Quality Management System. Individuals can pursue Lead Auditor or Internal Auditor training credentials that demonstrate professional competency in medical device quality systems.
How much do ISO 13485 consultants or auditors charge?
Consultant fees range from $5,000–$30,000 for small companies to $50,000–$100,000+ for large organizations. Certification body audit fees run $10,000–$20,000 for initial certification and $2,000–$15,000 for annual surveillance. Medical device specialists typically cost 20–40% more than generalists but achieve higher first-pass audit rates.


