
Introduction
IQ, OQ, PQ documentation is a structured, three-phase qualification process used in FDA-regulated industries to demonstrate that manufacturing equipment and systems are properly installed, operating correctly, and consistently performing as intended.
This framework is essential for quality assurance managers, regulatory affairs professionals, and medical device or pharmaceutical manufacturers building or auditing their qualification packages for FDA compliance.
The stakes are high: inadequate documentation is one of the most common triggers for FDA Form 483 observations and audit failures. Recent FDA warning letters issued to Bio-Medical Pharmaceutical Manufacturing Corporation (February 2026) and Medical Products Laboratories, Inc. (April 2026) explicitly cite failures to validate processes and qualify equipment as significant CGMP violations.
This article explains what each qualification phase involves, what FDA requires in writing, and what documentation errors to avoid so you can build audit-ready qualification packages that withstand regulatory scrutiny.
TL;DR
- IQ documents that equipment was delivered, installed, and configured per manufacturer specifications
- OQ confirms equipment operates correctly across all defined parameters and expected conditions
- PQ proves the process consistently produces acceptable results under actual production conditions
- All three phases must occur sequentially, with formal protocol, execution records, and summary report to meet FDA expectations
- Incomplete or poorly structured IQ/OQ/PQ documentation is a leading cause of FDA audit observations in medical device and pharmaceutical manufacturing
What Is IQ, OQ, PQ Documentation?
IQ/OQ/PQ is a three-stage qualification framework used to validate that equipment and manufacturing processes will consistently produce products meeting predetermined quality requirements. This is not a checkbox exercise but a documented evidence trail that demonstrates compliance with regulatory mandates.
Qualification vs. Validation: Understanding the Distinction
These terms have distinct meanings under FDA's framework:
- Qualification proves equipment is fit for its intended purpose
- Validation proves a process consistently produces a defined outcome
IQ/OQ/PQ serves as the foundational documentation layer for process validation under FDA's lifecycle approach. According to FDA's 2011 Process Validation Guidance, qualification demonstrates that utilities and equipment are suitable for intended use and perform properly, while validation addresses the broader manufacturing process across its lifecycle.
The Four-Stage Qualification Lifecycle
Many organizations include a precursor step called Design Qualification (DQ), which verifies the proposed equipment design meets user requirements before installation begins. According to EU GMP Annex 15, DQ is "documented verification that the proposed design of facilities, systems, and equipment is suitable for the intended purpose."
The full lifecycle progresses through four sequential stages, each building the evidentiary foundation for the next:
- DQ — Verifies the equipment design meets user and regulatory requirements
- IQ — Confirms correct installation per manufacturer specifications
- OQ — Demonstrates equipment operates within defined parameters
- PQ — Proves the process consistently produces product meeting acceptance criteria
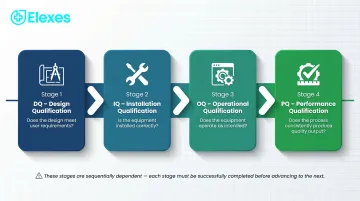
Together, these stages create a qualification record that withstands FDA inspection and audit scrutiny.
Why IQ, OQ, PQ Documentation Is Critical for FDA Compliance
The Regulatory Mandate
IQ/OQ/PQ documentation is not a best practice—it's a regulatory requirement:
For Medical Devices: As of February 2024, FDA updated 21 CFR Part 820 to the Quality Management System Regulation (QMSR), incorporating ISO 13485:2016 by reference. Equipment qualification and process validation expectations are now addressed through ISO 13485 clause 7.5.6, which requires validation of processes where the resulting output cannot be verified by subsequent monitoring or measurement.
For Pharmaceuticals and Biologics: Current Good Manufacturing Practice (CGMP) regulations under 21 CFR Parts 210/211 explicitly require manufacturers to document evidence of equipment qualification before commercial production. Sections frequently cited in recent FDA warning letters include:
- 21 CFR 211.100(a) — production and process controls
- 21 CFR 211.113(b) — aseptic and sterilization process validation
- 21 CFR 211.63 — equipment design, size, and location
Real-World Consequences of Documentation Gaps
When IQ/OQ/PQ documentation is inadequate or missing, companies face serious consequences:
- Product inconsistency due to unvalidated equipment
- Inability to investigate deviations when problems arise
- Exposure during FDA inspections leading to warning letters
Recent warning letters issued by FDA cite inadequate qualification of equipment, incomplete validation studies, and reliance on unvalidated processes as major CGMP violations. These citations frequently result in product holds, consent decrees, and costly remediation efforts that can take months to resolve.
Global Market Access
The documentation burden doesn't end with FDA. IQ/OQ/PQ protocols directly support ISO 13485 compliance, which means they're equally relevant for international market access. Companies targeting CE marking under EU MDR or registrations in Canada (Health Canada) and Australia (TGA) must demonstrate equivalent qualification documentation to satisfy regulatory requirements across multiple markets.
How Each Qualification Phase Works
The three phases must run in sequence—IQ cannot be skipped to reach OQ, and PQ cannot begin until OQ is complete—because each phase builds on confirmed outputs of the previous one.
Installation Qualification (IQ)
Core Question: Has everything been installed correctly?
IQ confirms the equipment arrived undamaged, was installed in the correct location, connected to utilities, and configured to manufacturer specifications.
What IQ Verifies:
- Physical installation checks (location, environment, access)
- Environmental conditions (temperature, humidity, cleanliness)
- Power supply and utility connections
- Calibration records and instrument verification
- Software version confirmation and configuration
- Documentation of manuals, certificates, and spare parts inventory
According to EU GMP Annex 15, IQ should include verification of correct installation against drawings and specifications, collection of supplier operating and maintenance instructions, calibration of instrumentation, and verification of materials of construction.
IQ Documentation Package:
- IQ Protocol: defines scope, methodology, and acceptance criteria
- IQ Checklist: derived directly from manufacturer requirements
- IQ Report: records findings and documents pass/fail determination
Note that CGMP requires the methodology to also be referenced in the Validation Master Plan (VMP), providing traceability to the organization's overall validation strategy.
Operational Qualification (OQ)
Core Question: Is the equipment operating correctly within its specified range?
OQ involves testing all operational functions at both nominal and worst-case operating limits—not just standard conditions. This phase confirms the equipment performs as designed across the full range of anticipated use.
What OQ Tests:
- Temperature controls and uniformity
- Pressure and vacuum controllers
- Humidity measurement and control
- Alarms and alert systems
- Interlocks and safety systems
- Automated processes and programming
- Data collection and reporting accuracy
EU GMP Annex 15 requires OQ to include tests confirming upper and lower operating limits and worst-case conditions to demonstrate equipment robustness.
OQ Documentation Package:
- OQ Protocol: objectives, methodology, and acceptance criteria
- OQ Test Scripts or Checklists: step-by-step procedures for each test
- OQ Report: records results, deviations, and conclusions
- Supporting Documentation: SOPs and calibration records
- Traceability Matrix: links each test back to user requirement specifications (URS)

Performance Qualification (PQ)
Unlike IQ and OQ, which test components individually, PQ tests the entire system together under real production conditions. It runs in the actual facility with trained personnel, using production materials, utilities, and procedures—the closest simulation of day-to-day operations before commercial manufacturing begins.
What PQ Demonstrates:
- Process capability and reproducibility
- Product quality consistency within and between batches
- System performance under full production load
- Operator capability and training effectiveness
- Integration of all equipment, materials, and procedures
FDA's 2011 Process Validation Guidance specifies what a written PQ protocol must cover. Required elements fall into two categories:
Process and testing requirements:
- Manufacturing conditions: operating parameters, processing limits, and component inputs
- Data collection and evaluation plan
- Acceptance criteria for each significant processing step
- Sampling plan covering within-batch and between-batch quality
Documentation and readiness requirements:
- Analysis methodology supporting science- and risk-based decisions
- Confirmed qualification status of all facility and equipment
- Personnel training and qualification records
- Material sources and analytical method validation status
Critical Requirement: The Quality Unit must formally approve the PQ protocol before execution begins. FDA explicitly requires this review and sign-off—starting PQ without it puts the entire validation at risk.
Essential IQ, OQ, PQ Documentation Requirements for FDA Compliance
Validation Master Plan (VMP)
The VMP is the overarching document that maps all qualification and validation activities across the organization. While FDA's 2011 guidance does not mandate the term "Validation Master Plan," EU GMP Annex 15 requires key elements of the site qualification and validation program to be defined and documented in a VMP or equivalent document.
VMP Contents:
- Validation policy and organizational roles
- Site inventory and qualification status
- Change control and deviation management procedures
- Guidance on acceptance criteria
- Document references and version control
- Strategy including requalification triggers
During FDA inspections, investigators commonly use the VMP to identify gaps between documented validation commitments and actual practice — particularly missing requalification triggers and undefined change control thresholds.
Three-Document Structure for Each Phase
Each qualification phase requires a standard documentation set:
- Protocol — pre-approved document defining scope, methodology, and acceptance criteria
- Execution Records — checklists and test scripts with real-time data entry
- Summary Report — final analysis, deviation resolution, and pass/fail conclusion
Critical Rule: Pre-approved protocols with clearly defined acceptance criteria are required before testing begins. Writing acceptance criteria after the fact constitutes a data integrity violation under FDA's 21 CFR Part 11 expectations.
Deviation Handling Documentation
When execution records reveal results outside acceptance criteria, those findings flow directly into deviation handling — the mechanism that determines whether a qualification phase can proceed or must be repeated. Any out-of-spec result must be formally documented, assessed for impact, and resolved through Corrective and Preventive Action (CAPA) before the qualification is considered complete.
According to EU GMP Annex 15, organizations must:
- Document and scientifically justify any significant protocol changes as deviations
- Investigate results failing to meet predefined acceptance criteria
- Address implications before progressing to next stages
Unresolved deviations cannot be dismissed in the final report. Each requires a formal investigation, root cause analysis, and documented corrective action before the summary report can be approved.
User Requirement Specifications (URS) and Traceability
Every test in OQ and PQ must be traceable back to a documented user requirement or regulatory requirement through a traceability matrix. Inspectors use this matrix to verify that every test serves a defined purpose — not just that testing occurred, but that it covered all critical requirements.
Traceability Best Practices:
- Document all user requirements before equipment selection
- Link each IQ check, OQ test, and PQ assessment to specific requirements
- Maintain version control throughout the qualification lifecycle
- Update traceability matrices when requirements or tests change
Re-Qualification Documentation Triggers
Equipment that undergoes significant modification, relocation, or major maintenance requires re-qualification documentation. FDA's 2011 guidance calls for periodic assessment of equipment and facility qualification data to determine whether requalification should be performed and to what extent.
Common Re-Qualification Triggers:
- Equipment relocation to a different facility or room
- Major component replacement or system upgrades
- Changes to critical operating parameters
- Significant process changes or new product introductions
- Periodic re-qualification intervals (time-based)
- Investigation findings following product quality issues
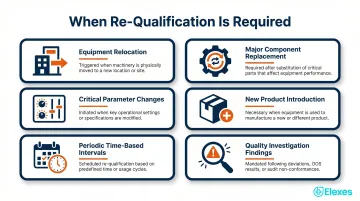
The written procedure should specify not just which triggers apply, but the scope required for each — whether a targeted OQ is sufficient or whether full IQ/OQ/PQ must be repeated from the start.
Common IQ, OQ, PQ Documentation Mistakes That Trigger FDA Findings
Mistake 1: Vague or Absent Acceptance Criteria
Stating "equipment shall be installed per manufacturer's instructions" is not measurable and won't satisfy an FDA reviewer. Acceptance criteria must be:
- Specific and quantifiable
- Objectively measurable
- Pre-approved before testing begins
- Documented with clear pass/fail boundaries
Compliant: "Autoclave chamber temperature shall maintain 121°C ± 2°C for the entire sterilization cycle duration."
Non-compliant: "Autoclave shall operate properly."
Vague criteria are easy to fix before testing begins — but the next mistake tends to surface only when companies try to close out their validation package.
Mistake 2: Conflating OQ and PQ
Many companies treat OQ and PQ as interchangeable, or use one to substitute for the other. They're not the same. Here's where the distinction matters:
| Aspect | OQ | PQ |
|--------|----|----|
| What it tests | Equipment function in isolation | Full process with all variables |
| Materials used | Simulated or surrogate materials | Actual production materials |
| Conditions | Controlled laboratory conditions | Real operating environment |
| Personnel | Validation team or engineers | Trained production operators |
| Objective | Prove equipment capability | Prove process reproducibility |
Conflating the two phases leaves a compliance gap FDA will flag. Both phases require distinct protocols and separately documented evidence — one cannot substitute for the other.
When OQ and PQ protocols are in order, the harder challenge is often something more fundamental: keeping documentation honest as execution happens.
Mistake 3: Retroactive Documentation
Protocols written or modified after testing — or reports signed without real-time data capture — are data integrity violations under 21 CFR Part 11 and CGMP. FDA's Data Integrity Guidance (December 2018) requires all records to meet ALCOA standards:
- Attributable — who performed the activity and when
- Legible — clear, permanent, and readable
- Contemporaneous — recorded in real time as the activity occurs
- Original — first recording or certified true copy
- Accurate — correct, truthful, and complete

Retroactive recordkeeping is one of the most frequently cited data integrity violations in FDA warning letters. Inspectors are trained to spot it.
Red flags FDA looks for:
- Protocol approval dates after test execution dates
- Test data recorded on loose sheets and transcribed later
- Identical handwriting styles across different test dates
- Missing or incomplete real-time data capture
- Electronic timestamps that don't align with reported activities
Conclusion
IQ, OQ, PQ documentation is the evidentiary foundation for demonstrating FDA compliance—each phase produces a structured, traceable record proving that equipment is installed correctly, operates within specification, and consistently delivers quality outcomes. Getting the documentation right matters as much as running the tests — inspectors scrutinize both.
Companies navigating IQ/OQ/PQ for the first time — or closing documentation gaps before an FDA inspection — benefit from consultants who know both the technical requirements and the standard FDA expects on paper. Elexes' QMS support services guide medical device and pharmaceutical teams through qualification documentation, from protocol development to final report approval.
With 256+ global clients and a 90% audit clearance rate across 250+ successful projects, Elexes brings proven experience preparing manufacturers for FDA, MDSAP, and Notified Body audits.
Frequently Asked Questions
What is IQ OQ documentation?
IQ OQ documentation refers to the formal written evidence produced during Installation Qualification and Operational Qualification—including protocols, test scripts, and summary reports—that proves equipment was installed and is operating correctly per manufacturer specifications and regulatory requirements.
What are the 4 stages of qualification?
The four stages are Design Qualification (DQ), Installation Qualification (IQ), Operational Qualification (OQ), and Performance Qualification (PQ). Each stage builds on the previous one, forming the complete qualification lifecycle required for FDA-regulated manufacturing.
What's the difference between OQ and PQ?
OQ tests whether equipment operates correctly within its defined limits, typically using simulated conditions or surrogate materials. PQ goes further, confirming the entire process consistently produces acceptable results using actual production materials, personnel, and real facility conditions.
What is IQ, OQ, and PQ in computer system validation?
In computer system validation (CSV), IQ verifies software is correctly installed and configured, OQ tests that system functions perform as specified within defined parameters, and PQ confirms the system consistently supports intended business processes under real operational conditions. FDA has also introduced Computer Software Assurance (CSA) as a risk-based alternative approach.
What are DQ documents?
DQ documents verify that proposed equipment or system design meets all user and regulatory requirements before procurement or installation begins. They typically include a User Requirement Specification (URS), design specifications, and a formal review confirming fitness for purpose.
Is PQ the same as UAT?
No. UAT is an IT concept focused on confirming user needs are met, while PQ is a formal GxP-regulated activity requiring pre-approved protocols, defined acceptance criteria, and a Quality Unit-reviewed summary report. The regulatory obligations governing PQ have no equivalent in standard UAT practice.


