Global Standards for Wearable Medical Devices: From Design to Market
Wearable medical devices have emerged as a ground-breaking technology, revolutionizing healthcare and empowering individuals to actively manage their health. These portable and smart devices, ranging from fitness trackers to advanced medical-grade wearables, offer real-time monitoring and health insights, enhancing daily living and disease management. However, with this rapid growth, it is of utmost importance to ensure that these devices meet stringent international standards and regulations.
It is important to understand that there are several different kinds of wearable devices but here, in this article, we are mostly covering the regulations aspect for wearable medical devices. To know the difference between wearable device and wearable medical device, you can contact our experts at [email protected].
Elexes has helped wearable device companies navigate ISO, FDA, and MDR regulations across 15+ product launches. If you’re building a wearable medical device, this guide simplifies what global standards you need to comply with, right from prototyping to market clearance.
What are Wearable Devices in Healthcare?
Wearable medical devices are compact and portable electronic devices, that are designed to be worn on the body to monitor various health-related parameters, collect data, and provide valuable insights to users and healthcare professionals.
These devices have gained significant popularity due to their –
Wearable technologies in Health Care assist in tracking various physiological parameters and health metrics, providing valuable information for both patients and healthcare professionals.
Various Purposes of Wearable Medical Devices
Patient Health Monitoring
Wearable devices can track vital signs like heart rate, blood pressure, respiratory rate, body temperature, activity levels, sleep patterns, and stress levels.
Leading smartwatch brands, including Apple, Samsung, Fitbit (now owned by Google), and Garmin, have been providing advanced features.
Chronic Disease Management
Certain devices are specialized for managing chronic conditions like diabetes by monitoring blood glucose levels continuously, allowing users to make informed decisions about their diet and medication.
Several companies like Dexcom, Abbott's FreeStyle Libre, and Medtronic have been at the forefront of Continuous Glucose Monitoring (CGM) technology.
Fitness Tracking
These popular devices cater to fitness enthusiasts by monitoring steps, distance covered, calories burned, and other exercise-related metrics and often sync with smartphone apps.
Companies like Fitbit, Garmin, and Xiaomi have continued to refine their fitness tracker offerings.
Early Detection and Prevention
Wearable devices with advanced sensors detect early signs of certain health conditions, allowing for timely intervention and prevention.
Portable ECG devices, like the AliveCor KardiaMobile, allow users to record their electrocardiogram and detect irregular heart rhythms, potentially aiding in the early detection of heart-related issues.
The Escalating Demand for Wearable Technologies
The growing market for wearable technologies in health care has witnessed a surge in popularity. These wearables have revolutionized the way individuals manage their health, providing real-time health insights and improving overall well-being.
Factors Influencing the Market Growth and Demand
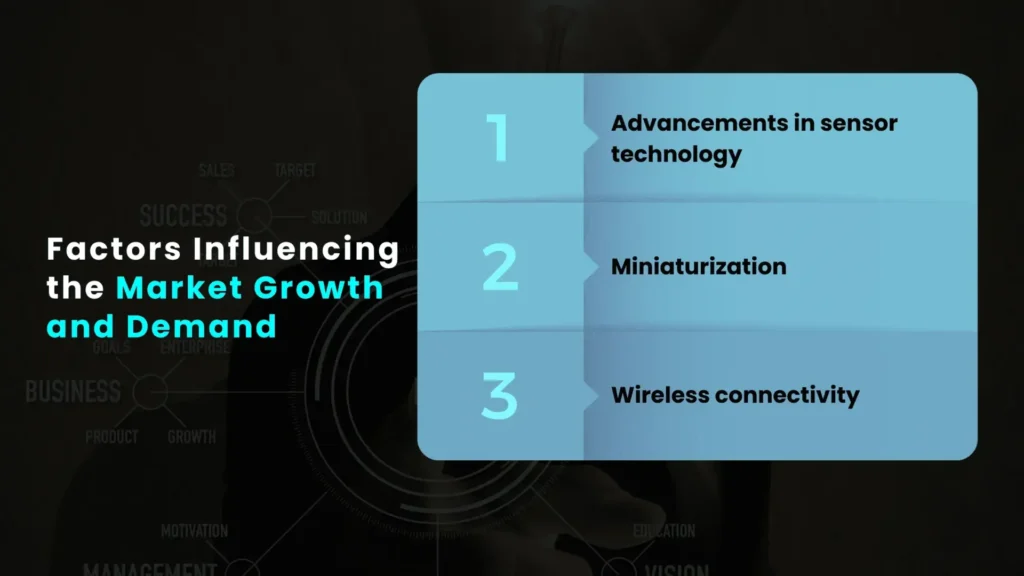
The market’s growth is fuelled by
With the integration of wearable medical technologies into digital health platforms and mobile apps, users can access personalized insights and recommendations based on data collected from their wearables.
Moreover, the seamless integration of wearables with the Internet of Things (IoT) and Artificial Intelligence (AI) has facilitated predictive modeling, enhancing data exchange and communication between devices.
However, the market still faces challenges related to data privacy, sensor accuracy, and the seamless integration of wearables into existing healthcare systems.
Despite these challenges, the market continues to evolve, presenting promising opportunities for personalized healthcare and empowering individuals to take a more active role in their health and well-being.
Obstacles Associated with Wearable Medical Devices

Wearable technology in healthcare has gained significant popularity and offers various benefits, but it also comes with its share of challenges. Let’s see what are the challenges that manufacturers are facing worldwide with wearable technologies in healthcare.
1. Data Accuracy and Reliability
Patient safety may be compromised due to inaccurate results leading to incorrect diagnoses or decisions by the healthcare provider. It is essential that the data collected by the device is accurate and reliable.
2. Compliance with Regulations and Standards
To ensure that wearable technology in healthcare is safe, device manufacturers should navigate through complex regulatory processes and obtain approvals from health authorities, which can be time-consuming and costly.
3. Data Privacy and Security
As these devices collect sensitive data, data privacy and security are paramount. This data should be protected from hackers, trying to breach it and obtain unauthorized access to patient information. This is essential to maintaining patient trust and adhering to the regulations of data protection.
4. Interoperability and Integration
Integrating wearable medical devices with existing healthcare systems and electronic health records can be challenging due to differing data formats and standards. Ensuring seamless data exchange and interoperability is critical for efficient healthcare workflows.
5. User Compliance and Adoption
Patient adherence to wearing and using the devices regularly is vital for accurate and continuous data collection. Convincing patients to adopt wearable devices and comply with recommended usage can be a significant challenge.
6. Battery Life and Maintenance
Prolonged battery life is essential for uninterrupted data collection. Also, ongoing maintenance, such as calibration for the sensor or updates to the firmware, uses a lot of battery power and therefore poses a challenge in extending the battery life while supporting technical functionalities.
Bringing Wearable Medical Devices to the Market
Bringing wearable medical devices to the market involves a comprehensive and well-organized process. Here are the key steps to consider:
1. Market Research and Concept Development
Conduct thorough market research to understand –
Based on research, develop a clear and innovative concept considering things like the form factor, functionality, user experience, and integration with existing healthcare systems.
2. Regulatory Compliance
Understand the regulatory landscape in the target markets to ensure the device meets the required safety and quality standards, and complies with relevant regulations.
3. Design and Development
Employ a rigorous design and development process that adheres to the applicable international standards (e.g., ISO 13485, ISO 14971) and incorporates usability engineering principles to create a user-friendly and safe device.
4. Prototyping and Testing
Create prototypes and conduct comprehensive testing to verify the device’s performance, reliability, and safety. Involve real users in usability testing to gather valuable feedback for improvements.
5. Clinical Trials (if applicable)
For higher-risk wearable medical devices, conduct clinical trials to gather scientific evidence supporting the device’s safety and efficacy.
6. Manufacturing and Quality Control
Establish a reliable manufacturing process and implement robust quality control measures to ensure consistency and compliance with specifications.
7. Regulatory Submissions
Prepare and submit the necessary regulatory documentation/applications for clearance or approval. Work closely with regulatory experts to navigate the submission process smoothly.
You can check out the most common mistakes that people do while 510k submission and avoid them.
8. Marketing and Distribution Strategy
Develop a sales strategy to promote the device to healthcare professionals, consumers, and potential distribution partners. Highlight its unique features and benefits. Consider online sales platforms, healthcare facilities, and specialized medical distributors.
9. Post-Market Surveillance
Implement a post-market surveillance system to monitor the device’s performance and safety in real-world use. Address any reported issues promptly.
10. Support and Continuous Improvement
Continuously support and gather user feedback and monitor market trends to identify opportunities for product improvement and new features.
11. Stay Updated with Regulations
Keep abreast of changing regulations and standards to ensure ongoing compliance throughout the device’s lifecycle.
What do US and EU regulations say about Wearable Devices?

Food and Drug Administration (FDA): In the U.S., wearable medical devices fall under the jurisdiction of the FDA, specifically the Center for Devices and Radiological Health (CDRH).
As with any other medical devices, medical devices with wearable technologies shall be classified based on their risk to patients and the level of control necessary to ensure their safety and effectiveness.
Risk Classification of medical devices in the US FDA is as follows:
You can also check out the details on medical device classification here in our post below:
The majority of Wearable Health Devices promote a healthy lifestyle and the FDA does have a compliance policy for such devices. Call us now to help you understand more about the policy.

Medical Devices Regulation (MDR): The EU has a regulatory framework for medical devices, and wearable medical devices fall under this regulation. The MDR aims to ensure the safety, performance, and quality of medical devices circulating within the EU market.
If you are struggling to identify the classification of your Wearable Health Device, book an appointment with us, We are happy to help.
What are the International Standards and Guidance to adhere to?
Several standards and guidance are relevant to wearable medical devices to ensure their safety, performance, and regulatory compliance.
These standards and guidance are established by international organizations and regulatory bodies to guide the development, testing, and certification/clearance of wearable health devices.
We should say, that the below is not an exhaustive list, as the standards and guidance can be added based on the features in the Wearable Health Devices.
Common standards/guidance applicable for all Wearable Medical Devices
| Standard Designation/Guidance | Standard Description | Purpose |
|---|---|---|
| IEC 60601-1 | General Requirements for Basic Safety and Essential Performance. | The device shall be tested to confirm that it met applicable standards of electrical safety. |
| ISO 14971 | Application of risk management to medical devices. | To identify and document the risks and hazards associated with the device and to implement the control measures to address the identified risks. |
| IEC 60601-1-2 | General requirements for basic safety and essential performance – Collateral Standard: Electromagnetic disturbances – Requirements and tests. | The device shall be tested to confirm that it met applicable standards for electromagnetic compatibility. |
| IEC 62366 | Application of usability engineering to medical devices. | The device shall be assessed with regard to Usability, as applicable. |
| IEC 62304 | Medical device software – Software life cycle processes. | To establish a structured framework to develop, verify and validate software to ensure safe and effective use of the software. |
| ISO 10993-1 | Biological evaluation of medical devices. | Testing and analysis of the device to demonstrate compliance with applicable standards of biocompatibility. |
| FDA Software Guidance | Content of Premarket Submissions for Device Software Functions. | To file software documentation as per the documentation level determined in accordance with the guidance. |
In case the device has a Wireless Radio Communication feature:
| Guidance | Guidance Description | Purpose |
|---|---|---|
| FDA Wireless Co-existence guidance. | FDA Radio Frequency Wireless Technology in Medical Devices Guidance issued August 2013. | The device shall be tested to ensure that device can communicate via wireless radio in its intended environment. |
Apart from this, in case there are any medical claims associated with the wearable medical device, clinical studies should be conducted to substantiate the claims and demonstrate equivalence to the predicate device.
Design Considerations (Parameters) in Designing Wearable Medical Devices
Designing wearable health devices requires careful consideration of various parameters to ensure their effectiveness, comfort, and safety. Some key design considerations for wearable medical devices include:
Ergonomics and Comfort
The device should be ergonomically designed to fit comfortably on the user’s body for extended periods. The choice of materials, weight distribution, and shape should minimize discomfort and skin irritation.
For example - in the design of a wearable insulin pump, the device needs to be worn consistently for effective glucose management. If the pump is not ergonomically designed, with consideration for factors like weight distribution and ease of movement, it could lead to discomfort and skin irritation for the wearer.
Sensor Accuracy and Reliability
Calibrated and validated sensors ensure that the device provides accurate measurements and data.
User Interface and Interaction
The design of the user interface must prioritize intuitiveness and ease of navigation, particularly for older individuals or those with physical limitations. The incorporation of user-friendly buttons, touchscreens, or voice commands should ensure accessibility for all users.
In the case of wearable heart rate monitors, a clear and intuitive display, along with user-friendly controls, enables individuals to easily access their heart rate data during various activities.
Connectivity and Data Transfer
Seamless connectivity to smartphones, tablets, or cloud platforms is vital for data transfer and analysis.
Data Security and Privacy
Wearable medical devices collect sensitive health data, necessitating robust encryption and data protection measures to safeguard user privacy and comply with data protection regulations.
Water and Dust Resistance
Many wearables are exposed to various environmental conditions, so they should be designed with appropriate water and dust resistance to ensure durability and longevity.
Adherence and Wearability
Design features that promote user adherence, such as easy on/off mechanisms, adjustable straps, or interchangeable accessories, can encourage consistent device usage.
We worked with a wearable respiratory monitor for patients with chronic lung conditions. The device needs to be water and dust-resistant, adhere comfortably to the skin, and be wearable throughout various activities. Our team collaborated with developers to ensure the selected appropriate materials, have appropriate durability and comfort as per applicable standard requirements. We guided the integration of specialized adhesives that maintain a secure fit even during physical exertion.
Power Management and Battery Life
Efficient power management is essential for wearable devices. Longer battery life or alternative power sources, such as solar or kinetic energy, can enhance device usability and reduce the need for frequent recharging.
Medical Grade and Regulatory Compliance
Depending on the device’s classification, manufacturers must adhere to relevant medical device regulations and standards, ensuring compliance with safety and performance requirements.
While using a wearable ECG monitor, patients need continuous heart monitoring, but frequent recharging can disrupt usage. While the company was struggling to understand regulatory expectations, Elexes collaborated with engineers to assist them in implementing power-saving features, like intelligent sleep modes and low-energy Bluetooth connectivity from a regulatory expertise standpoint.
Interoperability and Compatibility
Wearable devices may need to integrate with other medical devices or health platforms. Design considerations for interoperability and compatibility are essential for seamless data exchange.
Imagine a hospital that uses a comprehensive Patient Monitoring System to track vital signs and provide real-time data for patient care. The PMS consists of various interconnected medical devices, including Vital Sign Monitors to measure a patient’s vital signs, such as heart rate, blood pressure, and oxygen saturation, Central Monitoring Station to collect data from vital sign monitors and displays it in real-time for healthcare professionals to review, and Electronic Health Records (EHR) System to store patient records, including historical medical data and current vital signs.
What are the ramifications of not registering a wearable medical device?
Failing to register a wearable medical device can have serious consequences for both the manufacturer and the users of the device.
The ramifications of not registering a wearable medical device can vary depending on the country or region in which the manufacturer intends to market and sell the device.
Here are some potential ramifications:
Legal Consequences and Market Access Restrictions
Selling an unregistered medical device can lead to legal penalties, fines, or even criminal charges and devices shall be barred from entering the market.
Loss of Consumer Trust
Operating without proper registration can damage the reputation and consumers may lose trust in the device’s safety and effectiveness, impacting its sales and market acceptance..
Product Recall or Seizure and Patient Safety Risks
Regulators can recall products or seize unregistered medical devices if found on the market. This can cause financial losses and harm to users if the device is risky.
Competitive Disadvantage
Other registered products may have an advantage in terms of market access and credibility.
Conclusion
Wearable medical devices have ushered in a new era of personalized healthcare, empowering individuals to take control of their health. However, the successful development and market entry of these devices requires adherence to international standards, meticulous regulatory compliance, and a user-centric design approach. By prioritizing safety, accuracy, and patient privacy, wearable medical devices will continue to contribute significantly to advancing healthcare and improving the quality of life for millions of users worldwide.

How Elexes can help you bring your Wearable Medical Device to the Market?
Our team of expert medical device consultants is here to guide you every step of the way to bring your device to the market. With our in-depth knowledge of international regulations, standards, and best practices, we’ll ensure your device meets all necessary compliance requirements for smooth regulatory approval.
From navigating complex regulatory processes to conducting usability engineering studies and clinical trials, Elexes will streamline the development process and optimize your device’s performance and user experience. We’ll assist in designing robust risk management plans and quality management systems, guaranteeing the safety and effectiveness of your wearable medical device.
With our support, you can confidently address design considerations, including sensor accuracy, data security, and battery life, to create a user-friendly and reliable device that users will embrace. Whether you need assistance in competitive analysis, or regulatory or quality strategy, our team will help you make informed decisions for a successful market entry.
Don’t miss out on the opportunity to revolutionize healthcare with your wearable medical device.
Contact us today to get started on your journey to a successful wearable medical device launch.
Need Help Launching Your Wearable Device?
FAQs
Do wearables need CE or FDA approval?
Yes, if a wearable has a medical purpose and meets the definition of a medical device, it requires FDA clearance or CE marking before being marketed.
What ISO standards are applicable to wearable devices?
ISO 13485 for quality systems, ISO 14971 for risk management, IEC 62366 (usability engineering), IEC 60601-1 (electrical safety for active devices), ISO 10993-1 (biocompatibility for body contact) and IEC 62304 for software compliance are commonly applicable to wearable medical devices.
What makes a wearable a medical device?
A wearable becomes a medical device if it is intended to diagnose, monitor, prevent, or treat a disease or medical condition, as defined by regulatory authorities.
Is IEC 62304 mandatory for wearable software?
No, if the wearable includes embedded software or is considered Software as a Medical Device (SaMD), IEC 62304 is voluntary and often required by regulators.
Can wearables be classified as Class I devices?
Yes, Wearable medical devices may fall under Class I. However, diagnostic or therapeutic wearables often fall under Class II or III.






















