Medical Device Design & Development Guide – FDA, ISO & MDR Process
Imagine a world without medical devices: The lifesaving products that have revolutionized healthcare as we know it. From simple syringes to complex imaging machines, medical devices play a pivotal role in diagnosing, monitoring, and treating various medical conditions. But have you ever thought about the process behind the creation of these exceptional devices? Medical Device Design and Development is not just about inventing a new gadget; it is a structured, highly regulated process that ensures devices are safe, effective, and of the highest quality.
Elexes has guided 100+ MedTech firms through successful design control setup and Design History File (DHF) audits across FDA and MDR regions. Whether you’re preparing your DHF or launching a new device, understanding design controls is vital to de-risk timelines and ensure compliance.
What is a Medical Device?
Before delving into the intricacies of medical device design, let’s first understand what constitutes a medical device.
In simple terms, a medical device is any instrument, apparatus, machine, or implant used for medical purposes, be it for diagnosis, prevention, monitoring, or treatment of diseases.
These devices vary from basic tools like syringes to complex equipment like magnetic resonance imaging (MRI) machines. As medical technology advances and evolves, the need for innovative and highly effective medical devices grows in tandem.
The Art of Medical Device Design
Effective medical device design is paramount as it directly influences the device’s functionality, safety, and usability. Poorly designed medical devices can result in misdiagnoses, treatment errors, patient harm, or even fatalities.
Therefore, a well-thought-out medical device design process is crucial to ensure that devices are:
- User-friendly
- Accurate
- Reliable
- Compliant with regulations
But how do these designs take shape?
Like a sculptor chiseling away at a block of marble, medical device developers refine their concepts, ensuring each detail aligns with the desired outcome. The medical equipment design process is iterative, with constant feedback and improvements, akin to adding brushstrokes to a masterpiece.
Why is Medical Device Design Vital for Success in the Healthcare Landscape?
A medical device is a bridge connecting the realms of innovation and patient care, where the pivotal phase of medical device design and development acts as a backbone that determines the device’s success.
A well-crafted medical device not only complies with regulations but also delivers the intended functionalities, meeting market needs and gaining widespread adoption.
So, why is medical device design so crucial in the ever-evolving healthcare landscape?
Let’s explore…
Weaving Compliance and Innovation: The Regulatory Mandate
☛ The foundation of any medical device rests upon Quality and Regulatory Compliance, ensuring safety, efficacy, and user confidence
☛ A poorly designed medical device would lead to non-compliance and costly consequences
Quality and Regulatory Compliance are the bedrock of any medical device, ensuring safety, efficacy, and user confidence.
Delivering on Promises: Meeting Market Needs
☛ Medical devices serve as bridges between healthcare demands and technological advancements. Thoughtfully designed devices address specific medical needs, resonating with patients and healthcare professionals alike.
Embracing Creativity and Innovation
☛ Medical device design transcends concept generation, involving refined designs and creativity
☛ Expert developers invest significant time studying successful Medtech projects to craft cutting-edge solutions
Capturing the Essence: Developing Prototypes and Iterating
☛ Medical device developers create prototypes to test designs in real-world scenarios
☛ Iterative processes refine the device until it aligns perfectly with its intended purpose, eliminating compromise
Winning the Market Symphony: Gaining Market Adoption
☛ Well-designed medical devices gain market adoption and earn approval from healthcare providers, patients, and stakeholders
☛ Such devices become indispensable tools in healthcare, fostering innovation and elevating patient care standards
Empowering Healthcare Heroes: Enhancing Patient Outcomes
☛ Well-crafted medical devices equip healthcare professionals with advanced tools, resulting in exceptional patient care
☛Positive impacts on patient outcomes, diagnostics, and quality of life lead to significant improvements for countless individuals
Medical Device Design Control: A Step-by-Step Guide
The medical device design & development control process ensures that every aspect of device development, from inception to production, is thoughtfully considered and meticulously executed.
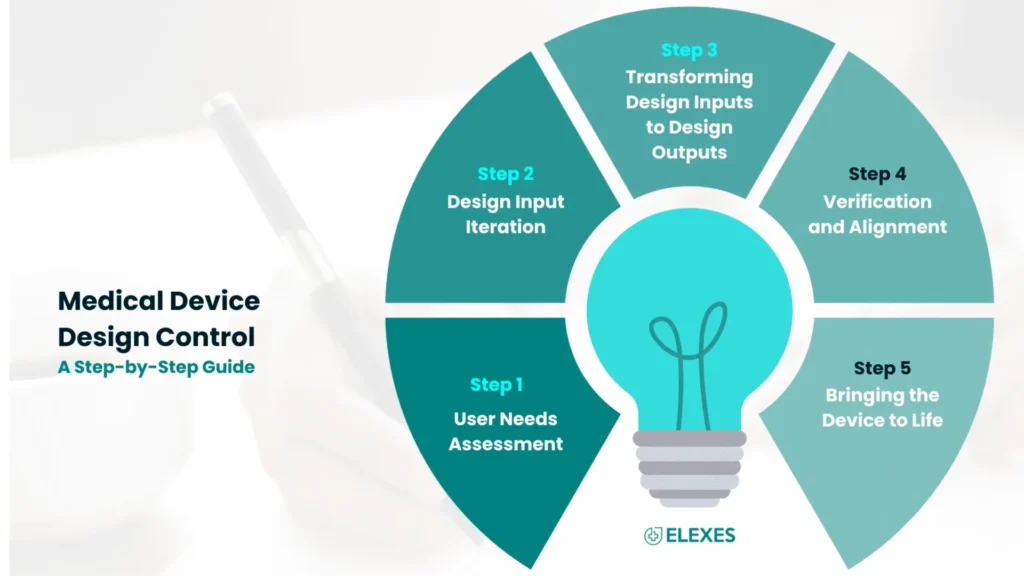
Step 1: Assessing User Needs
Before initiating the medical device design process, it’s crucial to thoroughly grasp both the market trends and the specific needs of potential users. Understanding the user needs will help you get a step ahead on understanding how you should proceed with medical device design & development process.
Step 2: Design Input Iteration
Building upon the insights gained from the user needs assessment, this step involves translating abstract user requirements into concrete design inputs. Through an iterative process, these inputs are refined and shaped.
Step 3: Transforming Design Inputs to Design Outputs
In this phase, the refined design inputs are converted into detailed design outputs. This involves specifying the technical details and functionalities of the device.
Step 4: Verification and Alignment
To ensure the design aligns perfectly with the original user needs and the established specifications, a thorough verification process is undertaken. This involves comprehensive testing, simulations, and evaluations to confirm that the design outputs meet the intended requirements.
Step 5: Bringing the Device to Life
With the verified and refined design in hand, the focus shifts toward mass manufacturing.
This step marks the transition from the design phase to the practical realization of the device, bringing it to life for the market and end-users.
Design control is not a linear process that concludes with the transfer to production. Instead, it remains an ongoing, dynamic endeavor. It continuously considers user feedback, usage patterns, and even the analysis of any previously failed devices.
Starting your medical device venture? Count us in as your expert guide. These steps are your blueprint for a product that ticks both tech boxes and user expectations. Email us now to get started.
Design Considerations for Medical Devices
Designers must ask critical questions to align their vision with customer demands and market needs.
• What the device is intended to do?
• What components are necessary for its success?
• Is there a plan for using it in conjunction with other devices?
• Are there specific performance targets to achieve?
• What potential risks might the end-users face?
• How can we improve upon existing products and address competitor weaknesses?
• What is the target cost per unit?
In diagnostic, therapeutic, and medical monitoring applications, three main considerations are pivotal in shaping medical device design.
Component and Space Optimization: On-patient wearables must prioritize compactness over the bulkiness often associated with more significant medical devices. Optimizing board space with newer micro-connectors and flexible circuitry accommodates the demand for smaller devices.
Flexible Circuitry Integration: Despite the shrinking size, modern medical devices are becoming more feature-rich. By employing flexible printed circuits (FPCs) and their corresponding cables and connectors, the circuitry integrated into the patient's body can achieve more precise and in-depth monitoring results.
Maximum Power and High Signal Integrity: Ensuring efficient powering of medical devices is crucial. Take into account the suitability of low-profile wire-to-board and flex-to-board options with the capacity to handle currents up to 15 amps. Board connections with higher circuit counts facilitate data transmission in compact form factors.
The Importance of Risk Management in Medical Device Design and Development
Risk management is a critical component of the medical device design and development process. Therefore, ensuring their safety and efficacy is of utmost importance.
Here’s how risk management plays a vital role in this process:
How Risk Management Works in Medical Device Design and Development?
Risk management in medical device design and development follows a structured process to identify, assess, mitigate, and monitor potential risks. Here’s a breakdown of how it works:
Risk management in medical device design and development follows a structured process to identify, assess, mitigate, and monitor potential risks. Here’s a breakdown of how it works:
- Risk Identification: Identify potential hazards and risks associated with the medical device, considering its use and potential errors.
- Risk Assessment: Evaluate risks based on severity, probability, and detectability to prioritize them.
- Risk Mitigation: Develop strategies to minimize risks, including design changes, safety features, and user instructions.
- Risk Control: Integrate risk control measures into the device’s development, ensuring effective risk reduction.
- Risk Monitoring: Continuously monitor device performance and post-market data for emerging risks.
- Documentation: Thoroughly document all risk-related activities, assessments, and outcomes throughout the process.
Addressing Regulation and Compliance Needs
Adherence to both regional and international regulations is imperative to guarantee the safety, quality, and effectiveness of medical devices.
Let’s explore the key points MedTech professionals should consider to address regulation and compliance needs effectively.
IEC 60601-1: The Universal Standard
IEC 60601-1 sets safety and performance requirements for medical electrical equipment. It includes usability engineering evaluations and human factor considerations. The standard updates regularly for emerging medical technologies and is essential for MedTech professionals.
ISO 13485: Global Quality Management Standard
ISO 13485 is a widely adopted standard for the quality management of medical devices. These ensure that strict quality control measures are followed during design, development, and manufacturing. Conformity to these standards is crucial for guaranteeing the safety and effectiveness of medical devices.
IEC 62304: Software Devlopment Standard
IEC 62304 outlines the software lifecycle processes for medical software, from conception to retirement, emphasizing risk management, documentation, and testing. By following this standard, developers can create efficient and safe medical software that meets healthcare industry requirements.
ISO 14971 - World-Wide Risk Standard
ISO 14971 helps medical equipment manufacturers identify and manage risks associated with medical devices throughout their lifecycles. By emphasizing a systematic approach, the standard promotes the development of safe products that prioritize patient well-being.
Understanding Global Medical Device Regulations
Medical device regulations serve as a comprehensive framework that ensures the safety, efficacy, and quality of medical devices while also promoting innovation and harmonization within the healthcare industry.
With varying requirements established by regulatory authorities in different countries, it is important to carefully analyze and adapt to the unique regulatory landscape of each market you wish to enter.
Here is a comprehensive overview of key global medical device regulations and their essential characteristics:
Regulation
Market
Key Points
US Food and Drug Administration (FDA)
United States
– Regulatory authority for medical devices in the United States
– Requires premarket clearance or approval for market entry
– Focus on safety and effectiveness
EU MDR (Medical Device Regulation)
European Union
– Replaces Medical Device Directive (MDD)
– Stricter requirements for device classification, clinical evidence, post-market surveillance
– Comply with Unique Device Identification (UDI) system
Health Canada MDR
Canada
– Requires medical equipment manufacturers to be licensed
– Focus on safety, efficacy, and quality
– Requires Medical Device Establishment License (MDEL)
Therapeutic Goods Administration (TGA) Regulations
Australia
– Regulation of medical devices through TGA
– Risk-based classification system
– Conformity assessment and ARTG listing required
Pharmaceuticals and Medical Devices Agency (PMDA) Regulations
Japan
– Strict approval process based on risk
– Requires marketing approval before market entry
China Food and Drug Administration (CFDA, now NMPA) Regulations
China
– Classification, registration, and approval required
– Focus on safety and quality
Agência Nacional de Vigilância Sanitária (ANVISA) Regulations
Brazil
-Device registration and post-market surveillance
– Conformity assessment for registration
Navigating the complex world of medical device regulations requires close collaboration with regulatory affairs (RA) and quality assurance (QA) professionals. RA/QA teams can provide valuable insights into regional and international standards and help ensure that the medical device design & development meets all compliance requirements.
Read here now to understand the pros and cons of hiring a medical device consultant.
By carefully addressing these factors, you can design a medical device that not only meets regulatory requirements but also delivers safe and effective healthcare solutions to patients and healthcare professionals.
How Elexes can help?
At Elexes, we provide medical equipment manufacturers our valuable insights into regulatory requirements, industry standards, and best practices, helping to navigate the complex landscape of medical device regulations.
Our team offers guidance in conceptualizing and refining product ideas, assisting in feasibility studies, and developing comprehensive project plans for DHF creation. Our deep understanding of the healthcare sector aids in aligning the device’s functionality with clinical needs.
Additionally, we facilitate effective communication between cross-functional teams, fostering collaboration among engineers, designers, clinicians, and regulatory experts. By tapping into their wealth of experience, we contribute to creating safe, innovative, and compliant medical solutions.
Conclusion
Designing and developing medical devices is a delicate balance of innovation, quality, and compliance. Meeting the needs of patients and healthcare professionals requires a deep understanding of the design process, regulatory landscape, and risk management principles. By adhering to design controls, considering human factors, and integrating emerging technologies, medical device manufacturers can create ground-breaking solutions that revolutionize healthcare.
Call us today. We will discuss how we can help your engineers and be your QA team.
Need Help Setting Up Your Design Controls?
FAQs
What are the FDA's required design controls?
FDA design controls include design and development planning, design inputs, design outputs, design review (internal and external), design verification, design validation, transfer, and design changes, as defined in 21 CFR 820.30.
How do I structure a Design History File (DHF)?
A DHF should include the design plan, input/output documents, risk assessments, verification/validation reports, reviews, and change control records.
What standards guide the development of medical device software?
IEC 62304 is the primary international standard for medical device software lifecycle processes, and it is often used alongside ISO 13485, ISO 14971 for risk management, and FDA requirements.
What is the difference between design verification and validation?
Design verification ensures that design outputs meet input requirements. Design validation confirms that the final product meets user needs and intended use.
Ex: Verification is testing in the lab that a blood pressure monitor inflates and measures pressure accurately, while Validation is making sure patients and doctors can use it easily and it helps guide treatment as intended.Is risk management a part of design controls?
Yes. Risk management, as per ISO 14971, must be integrated throughout the design process to identify, control, and monitor risks associated with the medical device.























