
For medical device companies navigating PMA in 2026, the difference between approval and rejection often comes down to the quality of their consulting partner. This list covers the 10 most capable PMA consulting firms in the USA based on demonstrated Class III device expertise, regulatory depth, clinical trial capability, and proven FDA CDRH familiarity.
TL;DR
- PMA is the FDA's most rigorous device pathway, requiring clinical proof of safety and effectiveness with costs reaching $10M–$100M+ from concept to approval
- Not all regulatory consultants handle Class III work; verify PMA submission history, IDE management experience, and CDRH expertise before engaging
- The 10 firms listed range from boutique PMA specialists to global consultancies, each with different strengths across strategy, clinical execution, and post-approval work
- Elexes, MCRA, M Squared Associates, and Smith Associates offer full-lifecycle PMA consulting across the USA
- Match consultant strengths to your device type, development stage, and internal regulatory bandwidth
What Is PMA and Why Does It Require a Specialist Consultant?
Premarket Approval (PMA) is the FDA's most stringent premarket review process, defined by the agency as the evaluation of safety and effectiveness for Class III medical devices. Unlike 510(k) clearances that rely on substantial equivalence, PMA demands independent proof through valid scientific evidence — typically from controlled clinical investigations.
Class III devices include those that support or sustain life, are permanently implanted, or present unreasonable risk of illness or injury. Examples include pacemakers, cochlear implants, implantable cardioverter-defibrillators (ICDs), deep brain stimulators, and certain drug-eluting coronary stents. These devices account for approximately 5% of all medical devices entering the US market — the remaining 95% use the less demanding 510(k) pathway.
Why PMA Submissions Demand Specialized Expertise
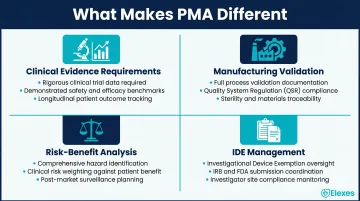
Four domains separate PMA from standard regulatory work:
- Clinical evidence requirements: PMA submissions must include data from pilot studies (20-100 patients) and pivotal trials (100-1,000+ patients), with appropriate control groups, statistical power calculations, and sufficient follow-up duration
- Manufacturing validation: Complete manufacturing process controls, validation protocols, and quality system documentation under 21 CFR Part 820
- Risk-benefit analysis: Comprehensive characterization of device risks, comparison to existing treatments, and documentation of mitigation strategies
- IDE management: Investigational Device Exemption applications to conduct clinical trials, including IRB coordination, informed consent documentation, and protocol amendments
The financial and timeline stakes are substantial. The FDA's statutory review target is 180 days from filing acceptance, but the FDA's actual performance runs significantly longer — 363.2 days on average in 2024, and 760.8 days in 2023.
Only 30-40% of PMA applications clear on the first review cycle, with each deficiency response costing $50,000-$300,000 in consulting and preparation fees.
The most common reasons for PMA delays or denials:
- Insufficient clinical evidence: underpowered studies, inappropriate control groups, inadequate follow-up duration
- Manufacturing deficiencies: inconsistent device performance, inadequate process validation, missing quality documentation
- Inadequate risk-benefit analysis: incomplete risk characterization, insufficient comparison to existing treatments
FDA user fees for FY2026 PMA submissions are $579,272 (standard rate) or $144,818 (small business rate) — and first-time applicants with gross receipts under $30M may qualify for a complete fee waiver. That fee, however, represents a small fraction of total PMA costs once consulting, clinical trials, and deficiency responses are factored in.
Generic regulatory consultants may handle 510(k) clearances competently but lack the clinical trial design experience, CDRH relationship management, and IDE support that Class III submissions require. Knowing what to look for in a PMA-specialist consultant — and where to find one — is where this guide starts.
Top 10 PMA Consultants in the USA for Medical Devices 2026
These firms were evaluated based on documented Class III device submission history, PMA-specific service offerings, clinical capabilities, quality system expertise, and market reputation in 2025–2026.
Elexes
Elexes is a globally recognized medical device regulatory and quality consulting firm with 50+ years of collective team experience, 250+ completed projects across 200+ product types, and a 90% audit clearance rate. The firm serves medical device companies, SaMD developers, IVD manufacturers, implant makers, and combination device companies worldwide — with notable clients including AliveCor, Outset Medical, and DJO Global.
PMA-specific strengths:
Elexes delivers end-to-end project management across the full submission lifecycle — from IDE support and clinical trial documentation through PMA preparation and post-approval surveillance. Their cross-functional outsourcing model brings regulatory, quality, and clinical expertise into a single engagement, with flexible staffing (full-time, part-time, or project-based) and complete data confidentiality.
The firm's Submission Readiness Assessment is built for PMA applications. It evaluates:
- Technical documentation completeness and traceability
- Clinical evidence adequacy against FDA CDRH review standards
- Risk management file integrity
- Quality system alignment across the full submission package
| Key PMA Services | PMA submission support, IDE preparation, clinical trial documentation, QMS compliance (ISO 13485, MDSAP), regulatory due diligence, post-approval support |
|---|---|
| Device Specialization | SaMD, IVD/LDT, implants, wearables, combination devices, companion diagnostics |
| Notable Differentiators | 50+ years collective experience, 256+ global clients, cross-functional outsourcing model, expertise across FDA, EU MDR, Health Canada, MHRA, and EMA |
MCRA (Medical Device Regulatory Advisors)
MCRA, headquartered in Washington, DC, is a full-service strategic consulting firm that integrates regulatory affairs, clinical trial management, and reimbursement planning. Now operating as an IQVIA business, MCRA maintains a strong track record in Class III device submissions and PMA applications.
Former FDA CDRH Branch Chief leadership and CMS advisors give MCRA a distinct edge in PMA strategy. The firm has managed IDE applications, breakthrough designation requests, and de novo petitions across neuromodulation, cardiovascular, and orthopedic segments. A published case study with ReGelTec documents IDE approval for the first pivotal US clinical trial for nucleus pulposus augmentation.
| Key PMA Services | PMA submissions, IDE, de novo, breakthrough designation, reimbursement strategy, clinical trial design |
|---|---|
| Device Specialization | Cardiovascular, orthopedics, neuromodulation, robotic-assisted surgery |
| Notable Differentiators | Former FDA CDRH Branch Chief leadership, former CMS advisors, integrated regulatory + reimbursement advisory, documented PMA track record |

M Squared Associates
M Squared Associates is a U.S.-based regulatory and clinical consulting firm specializing in medical devices and combination products reviewed by FDA CDRH. The firm offers full-service CRO capabilities alongside PMA submission expertise — a combination that allows for integrated regulatory and clinical management under one engagement.
M Squared manages multicenter pivotal studies supporting PMAs within defined budgets and timelines. The firm prepares both 510(k) and PMA submissions with an integrated quality and clinical approach, including CE Marking and Health Canada support for clients pursuing simultaneous market entry.
| Key PMA Services | PMA submissions, IDE support, pivotal clinical studies, QMS design (ISO 13485, 21 CFR Part 820), CE Marking |
|---|---|
| Device Specialization | CDRH-reviewed devices, combination products, IVDs |
| Notable Differentiators | Full-service CRO capabilities, U.S. and international clinical trial sites, integrated regulatory and clinical team |
Smith Associates (FDA Consultants)
Smith Associates is a regulatory consulting firm focused exclusively on medical devices, in vitro diagnostics, blood products, and biologics. The firm offers targeted PMA submission services, pre-submission meeting support, and Human Factors studies with a submission-focused approach.
Smith Associates takes a hands-on approach to PMA and IDE submissions, including Pre-IDE support and initial importer services. The firm builds practical, implementation-ready quality systems and supports manufacturers through 483 observation responses — a reliable option for companies seeking submission-specific expertise without large consulting overhead.
| Key PMA Services | PMA submissions, pre-submission meetings, IDE, Pre-IDE, Human Factors studies, U.S. Agent services |
|---|---|
| Device Specialization | Medical devices, IVDs, biologics, blood products |
| Notable Differentiators | Specialized FDA submission focus, practical implementation-ready quality systems, 483 observation response support |
Emergo by UL
Emergo by UL, headquartered in Austin, TX, is a global regulatory consulting firm offering market entry support in more than 20 countries. The firm provides deep expertise in FDA submissions, ISO 13485, MDSAP, and Software as a Medical Device (SaMD) regulatory pathways.
For PMA work, Emergo's integration with UL gives manufacturers access to biocompatibility evaluation resources and cybersecurity testing guidance — both critical when building the clinical and technical evidence package for Class III device approvals. The firm publishes detailed guidance on FDA cybersecurity expectations and ISO 10993 biocompatibility standards.
| Key PMA Services | FDA PMA/IDE support, QMS development, human factors engineering, UDI implementation, SaMD regulatory pathways |
|---|---|
| Device Specialization | SaMD, AI-driven devices, connected medical devices, Class II and Class III |
| Notable Differentiators | Global reach across 20+ countries, UL resources for testing guidance, strong cybersecurity and SaMD expertise |
NAMSA
NAMSA, headquartered in Northwood, OH, is a full-service medical device development organization with in-house laboratories for ISO 10993 biocompatibility testing, analytical chemistry, and preclinical/clinical study management. Founded in 1967, NAMSA operates 7 testing laboratories across the US and Europe.
NAMSA is well-suited for PMA submissions involving novel biomaterials or implantable devices. The firm manages the entire clinical and non-clinical evidence package — from bench testing through pivotal clinical trials — under one roof, reducing vendor coordination risk. NAMSA conducted 157,000+ medical device tests in the previous year with a 95% on-time turnaround rate.
| Key PMA Services | Biocompatibility testing (ISO 10993), clinical trial management, preclinical studies, FDA and EU MDR submissions |
|---|---|
| Device Specialization | Class III implantables, combination products, digital therapeutics, biomaterial-heavy devices |
| Notable Differentiators | In-house testing labs, end-to-end development-to-submission model, 13 Board-Certified DABT Toxicologists on staff |
MWA Consulting
MWA Consulting (recently acquired by AVS Life Sciences) is a GXP compliance consulting firm serving pharmaceutical, biotech, and medical device industries across the U.S., Canada, Europe, Asia, and South America. The firm employs over 300 experienced associates with broad capabilities across the full product lifecycle.
MWA's strength for PMA projects lies in its strong Clinical Quality Assurance (CQA) programs, GCP-compliant clinical trial infrastructure, and FDA mock inspection services. These capabilities help manufacturers verify that their clinical data and quality systems can withstand the scrutiny of a PMA review.
| Key PMA Services | GCP audits, clinical trial design and management, FDA compliance strategy, QMS (GMP/QSR), mock regulatory inspections |
|---|---|
| Device Specialization | Medical devices, pharmaceuticals, biotech combination products |
| Notable Differentiators | 300+ associates globally, standardized GXP training programs, cross-sector regulatory expertise |

Ken Block Consulting
Ken Block Consulting is a focused U.S.-based regulatory consultancy helping companies develop FDA-compliant strategies across the full medical device product lifecycle — from preliminary design activities through post-market surveillance.
The firm covers the full range of FDA submission types including PMA, 510(k), de novo, IDE, HUD, and Master Files. Ken Block Consulting is well-regarded for validation documentation support and quality system compliance with both 21 CFR Part 820 and ISO 13485 — making it a practical partner for manufacturers building PMA-ready quality infrastructure.
| Key PMA Services | PMA and IDE submissions, pre-market regulatory strategy, validation documentation, quality system compliance |
|---|---|
| Device Specialization | Broad medical device portfolio, U.S. and international markets (Canada, Europe, Australia) |
| Notable Differentiators | Full product lifecycle regulatory coverage, multi-submission type expertise, practical QMS implementation |
RQM+
RQM+, headquartered in Cary, NC, is a comprehensive regulatory and quality consulting firm with 40+ years of expertise and partnerships spanning 19 of the top 20 medical device companies and 7 of the top 10 IVD companies. The firm offers specialized services in biologics, combination products, and companion diagnostics alongside Class III device support.
RQM+ maintains strength in EU MDR/IVDR gap assessments and post-market clinical evaluation — increasingly relevant for manufacturers pursuing simultaneous FDA PMA and EU approval. This dual-pathway capability reduces duplication in clinical evidence generation and technical documentation, though specific PMA case studies are not publicly detailed.
| Key PMA Services | Regulatory consulting, EU MDR/IVDR submissions, clinical evaluation reports, post-market surveillance, laboratory testing (via Jordi Labs) |
|---|---|
| Device Specialization | Biologics, combination products, companion diagnostics, Class III devices |
| Notable Differentiators | 40+ years of expertise, partnerships with top MedTech companies, dual-pathway (FDA + EU) capability, ISO 9001 certified |
MDI Consultants
MDI Consultants is an established regulatory consulting firm with experience across medical devices, biotechnology, pharmaceuticals, and food companies. The firm helps clients achieve compliance across the U.S., Canada, and Europe, with particular strength in clinical trial management.
For PMA submissions, MDI offers end-to-end clinical trial management including protocol development, IRB interaction, IDE submission, trial monitoring, and statistical analysis. The firm also provides CE Marking support and FDA 483 crisis response — making it a well-rounded option for manufacturers managing complex PMA clinical study requirements. Mock FDA audits are performed by former US FDA field investigators.
| Key PMA Services | Clinical trial management (IDE, IRB, monitoring, statistical analysis), PMA submission support, FDA 483 response, CE Marking |
|---|---|
| Device Specialization | Medical devices, biotech, pharma, diagnostic devices |
| Notable Differentiators | Full clinical project management capabilities, multi-market compliance (U.S., Canada, EU), crisis intervention support |
How We Chose These Top PMA Consulting Firms
This list was compiled based on PMA-specific criteria — not general regulatory reputation. Many companies make the mistake of selecting consultants for PMA work based on brand name alone, without verifying Class III experience. The evaluation criteria used here directly address the documented reasons PMA submissions fail.
Class III Submission History and Clinical Trial Capability
We assessed each firm on:
- Demonstrated Class III device submission history (not just 510(k) experience)
- Ability to manage or support IDE and clinical trial programs
- FDA CDRH familiarity and track record of managing Pre-Sub meetings
- Quality system depth (ISO 13485, 21 CFR Part 820)
- Availability of post-approval support

Why this matters: Research shows insufficient clinical evidence is the leading cause of PMA delays and denials — including underpowered studies, inappropriate control groups, and insufficient follow-up duration. Manufacturing deficiencies and inadequate risk-benefit analysis are also major contributors.
Cross-Functional Integration and Practical Factors
We also evaluated each firm on practical factors that directly affect project outcomes:
- Flexibility in engagement model: Project-based vs. embedded support, scalability for startups vs. established manufacturers
- Cross-functional capabilities: Clinical + regulatory + quality expertise under one engagement (reduces vendor coordination risk)
- Responsiveness to FDA feedback: Track record of managing deficiency letters and interactive review
- Cost and timeline efficiency: Ability to proactively identify submission gaps, reduce rework cycles, and accelerate review timelines
The financial reality: PMA engagements typically span multiple years and represent significant financial exposure. Responding to FDA deficiency letters costs an estimated $50,000-$300,000 per cycle. Advisory panel preparation adds another $50,000-$200,000. Post-approval studies mandated by FDA carry a median cost of $2.16 million per study.
Conclusion
Choosing a PMA consultant is one of the most consequential decisions a Class III device manufacturer will make. The right partner not only increases the probability of FDA approval but actively manages risk, timelines, and costs throughout a multi-year regulatory journey.
The firms on this list represent a range of PMA consulting strengths:
- Elexes, MCRA, and M Squared Associates offer comprehensive end-to-end PMA support combining regulatory strategy, clinical execution, and quality system compliance
- NAMSA provides unique value for biomaterial-heavy implantables through in-house biocompatibility testing and preclinical study management
- Smith Associates and Ken Block Consulting deliver focused, submission-specific expertise without large consulting overhead
- Emergo by UL and RQM+ support simultaneous FDA and international market entry for global commercialization strategies
- MWA Consulting and MDI Consultants bring deep clinical trial management and GCP audit capabilities
Selecting the Right Partner for Your Device
Assess consultants based on:
- Device type and therapeutic category: confirm the consultant has Class III experience in your specific segment
- Development stage: determine whether you need full-spectrum IDE-through-post-approval support or submission preparation only
- Internal regulatory bandwidth: decide whether your team needs embedded expertise or periodic strategic guidance
Look beyond credentials to ask about:
- Relevant Class III submission case studies in your device category
- Typical PMA timelines and how the consultant manages FDA interactive review
- How they handle deficiency responses and amendment cycles
- Post-approval support availability for supplements and annual reports
Get Expert PMA Guidance from Elexes
Elexes offers flexible, project-based PMA consulting engagements with a team bringing 50+ years of collective experience across FDA, EU MDR, and global regulatory frameworks. The firm's Submission Readiness Assessment is specifically designed for PMA applications, identifying gaps and providing a prioritized action plan before filing.
Whether you're preparing your first Class III submission or managing a complex clinical trial program, Elexes provides the specialized expertise your PMA engagement requires. Contact Elexes today for an initial regulatory assessment and discover how end-to-end project management can accelerate your path to FDA approval.
Frequently Asked Questions
What is PMA consulting?
PMA consulting covers specialized regulatory advisory services that help medical device companies prepare and submit a Premarket Approval application to the FDA for Class III devices. Core services include clinical trial support, submission writing, IDE preparation, and FDA interaction management.
Is PMA legal for medical use?
PMA is not a substance or procedure — it's the FDA's formal approval designation for Class III medical devices. A device that has received PMA approval has been reviewed and authorized by the FDA as safe and effective for its intended medical use, making it fully legal to market and sell in the United States.
What types of medical devices require PMA approval?
Class III devices requiring PMA include those that sustain or support life, are permanently implanted in the body, or present unreasonable risk of illness or injury. Examples include pacemakers, cochlear implants, deep brain stimulators, implantable cardioverter-defibrillators (ICDs), replacement heart valves, and certain drug-eluting coronary stents.
How long does the FDA PMA review process take?
The FDA's statutory target is 180 days from filing acceptance, but actual timelines run longer — 363.2 days on average in 2024, and 760.8 days in 2023. From initial submission through amendments, advisory committee review, and final decision, total timelines commonly reach 12–36 months.
What is the difference between PMA and 510(k) submissions?
A 510(k) clears Class II devices by demonstrating substantial equivalence to a predicate and rarely requires clinical trials. A PMA requires independent proof of safety and effectiveness through controlled clinical evidence — making it far more demanding and costly ($10M–$100M+ vs. $100K–$500K for 510(k)).
How do I choose the right PMA consultant for my medical device?
Prioritize consultants with verifiable Class III device submission experience in your therapeutic category, clinical trial management capabilities, and a track record of managing FDA Pre-Sub meetings and interactive review. Ask for relevant case studies in your device category and confirm they offer engagement models suited to your stage and budget.


