
Introduction
February 2, 2026 marked a hard deadline for US medical device manufacturers: full compliance with the FDA's Quality Management System Regulation (QMSR), the most significant overhaul of domestic device quality requirements in decades. The regulation aligns US requirements with ISO 13485:2016 and introduces new documentation accessibility standards that fundamentally change how manufacturers prepare for inspections.
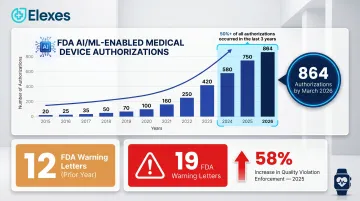
The enforcement pressure is real. FDA Warning Letters for quality system violations surged 58% in 2025, with 19 issued by early September compared to just 12 during the same period in 2024. CAPA deficiencies remain the most frequently cited observation during FDA inspections.
Post-market surveillance scrutiny has also intensified, particularly for Software as a Medical Device (SaMD) and AI/ML-enabled devices. These categories now account for over 50% of all device authorizations over the last three years.
The right quality consulting partner helps companies build defensible Quality Management Systems, navigate audits confidently, and reach market faster. The wrong choice can cost months of delays, remediation expenses exceeding $200,000, and lasting damage to compliance reputation.
This list profiles the top 10 medical device quality consulting firms serving the US market in 2026, selected for their QMS depth, audit performance, and proven client outcomes.
TLDR
- Medical device quality consulting firms build compliant QMS infrastructure aligned with FDA QMSR and ISO 13485 requirements
- The FDA QMSR transition and a 58% rise in quality-related Warning Letters make experienced quality partners essential in 2026
- Top firms were evaluated on QMS implementation expertise, audit clearance track records, team credentials, and engagement flexibility
- Elexes, Emergo by UL, RQM+, NAMSA, and RCA each lead in distinct areas — from startup support to global manufacturer compliance
- Shortlist 2-3 firms based on device class experience and assigned team qualifications
What Is Medical Device Quality Consulting — And Why It Matters in the US
Medical device quality consulting firms help manufacturers design, implement, maintain, and remediate Quality Management Systems that comply with FDA 21 CFR 820/QMSR, ISO 13485, and related international standards. These consultants prepare companies for regulatory inspections, support audit readiness, and build the QMS infrastructure that underpins successful market submissions.
The FDA's QMSR now incorporates ISO 13485:2016 by reference — a structural change that reshaped documentation obligations overnight. Manufacturers must now make internal audit reports, supplier audit reports, and management review records available to investigators — records previously exempt under the legacy QSR.
This creates direct demand for consulting firms that understand both ISO 13485 certification requirements and the additional FDA-specific obligations that go beyond the standard.
The global regulatory affairs outsourcing market reached $6.6 billion in 2023 and is projected to grow to $11.31 billion by 2030 at a CAGR of 8.3%, driven by increasing regulatory complexity and enforcement intensity. FDA enforcement data shows Warning Letters for device quality system violations increased 58% year-over-year in 2025 — a clear signal that quality system expertise is no longer a discretionary investment for manufacturers at any stage.
That enforcement pressure intersects with a separate but equally consequential trend: the FDA has authorized 864 AI/ML-enabled medical devices as of March 2026, with more than half of all historical authorizations occurring in the last three years. This acceleration has created specialized demand for consultants with expertise in software quality systems aligned with IEC 62304 and emerging AI/ML lifecycle management requirements.
Top 10 Medical Device Quality Consulting Firms in the US (2026)
These firms were selected based on QMS implementation depth, documented audit performance, team credentials (including RAC certification and ISO 13485 lead auditor qualifications), engagement model flexibility, and demonstrated experience across device classes and company stages.
Elexes Medical Consulting
Elexes is a globally active medical device regulatory and quality consulting firm with 50+ years of collective team experience, serving 100+ global clients across 200+ product types. The firm maintains a strong US client presence spanning startups to established manufacturers, with core quality services including ISO 13485 QMS implementation, FDA 21 CFR 820/QMSR compliance, MDSAP preparation, design controls, risk management (ISO 14971), clinical trial documentation, and post-market surveillance support.
Elexes differentiates through a cross-functional outsourcing model that combines regulatory, quality, and clinical tasks under unified engagements, eliminating the coordination overhead that typically slows multi-disciplinary projects. Flexible full-time, part-time, and project-based engagement models with complete data confidentiality make it accessible to both early-stage startups and scaling manufacturers targeting global markets.
| Core Quality Services | QMS implementation (ISO 13485/QMSR), MDSAP preparation, design controls, risk management, CAPA, post-market surveillance, clinical trial documentation |
|---|---|
| Key Standards & Certifications | ISO 13485, ISO 9001, ISO 14971, IEC 62304, MDSAP, GLP, GCP, HIPAA |
| Best For | Startups to global manufacturers needing end-to-end QMS and regulatory quality support across US, EU, and international markets |
Emergo by UL
Emergo by UL is one of the most established names in medical device quality and regulatory consulting, with 400+ professionals across 20+ global offices and deep expertise across US FDA, EU MDR, and international quality standards. Operating since 1997, the firm holds ISO 13485 certification and delivers services in English, Korean, Japanese, German, and Simplified Chinese to support multinational device manufacturers.
Core services span ISO 13485 implementation, FDA QMSR compliance, internal/supplier/mock FDA audits, MDSAP preparation, and full-lifecycle QMS support. As part of UL, Emergo combines regulatory consulting with product testing and certification capabilities in a single engagement, enabling integrated quality and testing workflows that reduce time-to-market. This model is particularly strong for mid-size to large manufacturers managing complex device portfolios across multiple jurisdictions.
| Core Quality Services | ISO 13485 QMS, FDA QMSR, internal/mock FDA audits, MDSAP, post-market surveillance |
|---|---|
| Key Standards & Certifications | ISO 13485, FDA 21 CFR 820/QMSR, MDSAP, EU MDR |
| Best For | Mid-size to large manufacturers seeking integrated quality consulting + testing from a single global partner |
RQM+
RQM+ is a pure-play life sciences regulatory and quality consulting firm formed through strategic mergers, with 400+ consultants and offices across the US, UK, Ireland, Germany, Netherlands, and Japan. The firm specializes in ISO 13485 implementation and remediation, FDA QMSR transition support, internal audits, mock FDA inspections, supplier audits, and MDSAP preparation.
A deep bench of former FDA officials and Notified Body professionals makes RQM+ exceptionally strong in quality system remediation, particularly for companies responding to FDA Warning Letters, 483 observations, or EU MDR nonconformities.
RQM+'s remediation practice focuses on building sustainable internal QMS capability rather than creating long-term consulting dependency — an approach that appeals to manufacturers seeking to strengthen in-house quality functions while addressing immediate compliance gaps.
| Core Quality Services | QMS remediation, ISO 13485, mock FDA inspections, MDSAP, CAPA management, supplier audits |
|---|---|
| Key Standards & Certifications | ISO 13485, FDA 21 CFR 820/QMSR, MDSAP, EU MDR/IVDR |
| Best For | Companies responding to FDA Warning Letters, preparing for inspections, or transitioning from QSR to QMSR |

NAMSA
NAMSA is a full-service Contract Research Organization combining regulatory quality consulting with extensive in-house laboratory testing capabilities, making it unique among firms offering both strategic QMS consulting and hands-on biocompatibility/sterilization testing. Founded in 1967, NAMSA serves more than 3,000 companies annually with a global team of 1,001–5,000 employees.
Core quality services include ISO 13485 compliance, design controls, risk management (ISO 14971), biocompatibility program support (ISO 10993), sterilization validation, and regulatory strategy.
The integrated CRO and quality model eliminates coordination friction for manufacturers of implantable or sterile devices, keeping quality documentation and test protocols aligned throughout development and reducing overall timelines.
| Core Quality Services | ISO 13485, design controls, risk management, biocompatibility planning, sterilization validation, QMS integration |
|---|---|
| Key Standards & Certifications | ISO 13485, ISO 14971, ISO 10993, FDA 21 CFR 820/QMSR |
| Best For | Manufacturers of implantable or sterile devices needing integrated quality consulting and laboratory testing |
Regulatory Compliance Associates (RCA)
RCA, a Nelson Labs company within the Sotera Health group, focuses exclusively on life sciences with a team that includes former FDA officials and industry veterans. The firm serves over 5,000 customers in 50+ countries, including 40 of the top 50 medical device companies, and holds ISO 9001 certification.
Core quality services include QMS development and implementation, FDA compliance remediation, ISO 13485 implementation, internal and mock FDA audits, CAPA management, and Warning Letter response support. RCA's depth in FDA compliance remediation makes it particularly valuable for US-based manufacturers navigating 483 observations, Warning Letters, or consent decrees. Its hands-on approach translates regulatory requirements into operational quality systems that sustain compliance well beyond initial certification.
| Core Quality Services | QMS development, FDA remediation, ISO 13485, CAPA, Warning Letter response, audit readiness |
|---|---|
| Key Standards & Certifications | ISO 13485, FDA 21 CFR 820/QMSR, 21 CFR Part 11 |
| Best For | US-based manufacturers needing FDA compliance remediation or building a QMS from scratch |
Medpoint
Medpoint is a full-service quality and regulatory consulting firm with 350 global consultants offering broad medical device quality services, including QA support for FDA 21 CFR 820/QMSR compliance, MDSAP readiness, clinical quality support, technical consulting, and global supplier auditing.
Its large team enables rapid scaling and multi-disciplinary support across quality, regulatory, clinical, and supplier management simultaneously, making it well-suited for companies managing several compliance workstreams at once. Medpoint's supplier audit capability, backed by an extensive global auditor network, delivers on-the-ground coverage across international supply chains — directly addressing one of the most persistent FDA citation areas: purchasing controls under 21 CFR 820.50.
| Core Quality Services | FDA 21 CFR 820/QMSR, MDSAP, supplier audits, clinical quality support, QMS maintenance |
|---|---|
| Key Standards & Certifications | ISO 13485, FDA QMSR, MDSAP, GMP standards |
| Best For | Companies needing scalable, multi-disciplinary quality support spanning QA, regulatory, and supplier management |

QES Medical
QES Medical is a Boston-based boutique consulting firm founded in 2009, specializing in quality and regulatory services for the medical device industry. Core services include QMS building and maintenance, FDA 21 CFR 820/QMSR compliance, ISO 13485 implementation, risk management (ISO 14971), design controls, audit readiness, and SaMD compliance (IEC 62304).
QES Medical is known for a hands-on approach to building QMS from the ground up, with the founder bringing 20+ years of leadership experience at Boston-area device firms. It's particularly effective for startups preparing for a first ISO 13485 certification or initial FDA inspection, offering guidance that scales to early-stage resource constraints without over-engineering the system.
| Core Quality Services | QMS build, ISO 13485, FDA QMSR, risk management, design controls, audit readiness, SaMD compliance |
|---|---|
| Key Standards & Certifications | ISO 13485, ISO 14971, IEC 62304, FDA 21 CFR 820/QMSR |
| Best For | Early-stage startups building their first QMS or preparing for initial ISO 13485 certification |
Rook Quality Systems
Rook Quality Systems, celebrating its 12th year, specializes in quality and regulatory strategy for emerging medical device companies, with 36 team members including 13 certified quality auditors. Services cover QMS development and maintenance, ISO 13485, FDA QMSR, MDSAP audits, EU MDR CE marking support, and SaMD/IVD quality compliance.
Rook focuses on lean quality system design for hardware, SaMD, and IVD companies across Class I–III, with strong capability in transitioning companies from paper-based to electronic QMS. Particular expertise in agile SaMD quality compliance (IEC 62304) and emerging AI/ML requirements positions it well for software-driven manufacturers navigating the January 2025 FDA draft guidance on AI-enabled device lifecycle management.
| Core Quality Services | QMS design, ISO 13485, MDSAP, SaMD quality (IEC 62304), EU MDR CE marking, eQMS transition |
|---|---|
| Key Standards & Certifications | ISO 13485, FDA 21 CFR 820/QMSR, MDSAP, IEC 62304, EU MDR |
| Best For | Emerging device companies, SaMD developers, and IVD manufacturers building efficient, audit-ready quality systems |
Cannon Quality Group
Cannon Quality Group (CQG), founded in 2010, is a full-service outsourced quality management company for medtech, IVDR, SaMD, and pharma companies. Core quality services include QMS design and implementation, document control, design control, audit management, risk management, CAPA, production control, complaint management, and supplier quality.
CQG focuses on "right-sized" quality systems that avoid over-burdening early-stage companies while remaining fully compliant with FDA and ISO requirements. Its outsourced quality model delivers fractional QA/QC support for startups and growth-stage companies that need expert oversight without the overhead of full-time hires, making it accessible to pre-revenue manufacturers preparing for first regulatory submissions.
| Core Quality Services | Outsourced QMS, document control, design control, CAPA, supplier audits, complaint management |
|---|---|
| Key Standards & Certifications | ISO 13485, ISO 14971, FDA 21 CFR 820, IEC 62366 |
| Best For | Medtech startups and growth-stage companies wanting fully outsourced, right-sized quality management |
MCRA
MCRA (Musculoskeletal Clinical Regulatory Advisers), now an IQVIA business, is a specialized consulting firm with deep expertise in musculoskeletal, orthopedic, cardiovascular, and spine devices. Quality services are integrated with regulatory and reimbursement capabilities, covering QMS compliance, clinical trial quality, FDA submission support, supplier audits, and cybersecurity quality requirements.
MCRA's specialized depth in orthopedic and musculoskeletal device quality is reinforced by a newly launched AI & Imaging Center led by former FDA imaging experts. A global network of surgeons supports streamlined clinical quality studies, and a strong track record with orthopedic De Novo applications makes it a natural fit for spine, joint reconstruction, and sports medicine manufacturers needing integrated quality, regulatory, and reimbursement strategy.
| Core Quality Services | QMS compliance, clinical quality, FDA submissions, cybersecurity quality, supplier audits |
|---|---|
| Key Standards & Certifications | ISO 13485, FDA 21 CFR 820/QMSR, IEC 62304 (SaMD/AI), ISO 14971 |
| Best For | Orthopedic, spine, cardiovascular, and musculoskeletal device manufacturers needing integrated quality + regulatory + reimbursement strategy |
How We Selected the Best Medical Device Quality Consulting Firms
These firms were evaluated on quality-specific expertise, verifiable audit outcomes, breadth of QMS services, and adaptability to companies at different stages. The most common mistake manufacturers make is selecting a firm based on name recognition alone—overlooking whether the assigned team (not the sales team) has direct experience with their device class or risk classification.
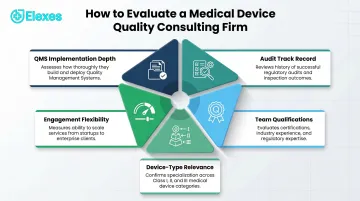
Key Selection Criteria:
- QMS Implementation Depth — Delivers documentation, procedures, and hands-on ISO 13485/FDA QMSR implementation—not just strategic guidance.
- Audit Track Record — CAPA is the #1 FDA 483 observation area, followed by complaint handling and process validation. Firms with documented remediation results in these areas ranked higher.
- Team Qualifications — RAC-Devices certification from RAPS and ISO 13485 lead auditor credentials are the leading competence indicators. Former FDA investigators or Notified Body professionals add regulatory perspective that industry-trained consultants alone cannot replicate.
- Engagement Flexibility — Project-based, retainer, and staff augmentation models serve different needs. Startups need fractional support; scaling companies need surge capacity during submissions; established manufacturers need ongoing compliance maintenance.
- Device-Type Relevance — Experience with your specific device class (active implantable, SaMD, IVD, combination product) and risk classification (Class I, II, III). A firm with strong orthopedic implant experience may lack the SaMD software lifecycle expertise required under IEC 62304.
Firms that provide detailed scopes of work, defined milestones, and clear deliverables produce better outcomes than those with vague proposals and open-ended billing structures.
Conclusion
The best medical device quality consulting firm for your company is not necessarily the largest or most recognized—it's the one with demonstrated experience in your device class, a team qualified to work on your specific QMS challenges, and an engagement model that fits your stage and budget.
A quality partner should build your internal capability over time, not create permanent dependency. When evaluating firms, look for:
- Transfers knowledge through documentation, not just deliverables
- Trains your team during implementation, not after
- Prepares you for independent audit readiness from day one
The goal is sustainable compliance, not ongoing consulting revenue.
Elexes brings 50+ years of collective experience and flexible engagement models ranging from project-based consulting to embedded quality professionals. The team supports medical device companies at every stage: QMS build-out for startups, MDSAP preparation, and post-market surveillance for global manufacturers. If you're evaluating quality consulting partners for 2026, reach out for a consultation to discuss your compliance objectives.
Frequently Asked Questions
What does a medical device quality consulting firm do?
Quality consultants help device companies design, implement, maintain, and remediate Quality Management Systems to comply with FDA 21 CFR 820/QMSR and ISO 13485. Core services include document control, CAPA systems, design controls, audit preparation, risk management (ISO 14971), and post-market surveillance.
When should a medical device company hire a quality consultant?
Common trigger points include:
- Early product development, to build quality-by-design into the process
- Pre-submission, to ensure QMS documentation supports technical files
- Before FDA inspections or ISO 13485 certification audits
- When responding to 483 observations or Warning Letters
What is the difference between regulatory consulting and quality consulting for medical devices?
Regulatory consulting focuses on submission strategy and market approval pathways (510(k), PMA, CE marking), while quality consulting focuses on the internal QMS infrastructure that supports those submissions—including design controls, document management, and CAPA systems. Many firms offer both, and the disciplines are deeply interconnected.
What certifications should a medical device quality consulting firm have?
Look for firms operating under ISO 13485 with consultants holding RAC-Devices (RAPS) and ISO 13485 lead auditor credentials. Familiarity with FDA QMSR, MDSAP, ISO 14971, and IEC 62304 is essential depending on your device type.
How much does medical device quality consulting typically cost?
Costs vary by scope and device complexity. ISO 13485 QMS implementation typically runs $25,000–$150,000 depending on company size and device class. Monthly retainers for ongoing support run $5,000–$30,000/month. Mock FDA inspections typically cost $10,000–$40,000.
How do I choose the right quality consulting firm for a medical device startup?
For early-stage companies, prioritize:
- Hands-on QMS build experience with startups, not just enterprise clients
- Flexible engagement models (retainer or project-based)
- Consultants who train internal staff and transfer knowledge, not just deliver documents
- Ability to right-size quality systems to your resources while maintaining full compliance


