
Introduction
Medical device manufacturers face mounting regulatory pressure from every direction. Between FDA's shift to the new QMSR (Quality Management System Regulation), stricter EU MDR enforcement, and the global expansion of MDSAP audits, compliance complexity has never been higher. A single failed FDA inspection can trigger delays that cost months of market time, while major recalls average $10M to $600M depending on scope and downstream impact.
Managing that risk starts with the right infrastructure. Your Quality Management System platform is the operational backbone that documents, controls, and demonstrates compliance across every stage of the device lifecycle — and with ISO 13485, FDA 21 CFR Part 820/QMSR, EU MDR, and MDSAP converging worldwide, the stakes of choosing the wrong one are real.
The wrong platform can derail regulatory timelines, inflate overhead costs, and block market access across key jurisdictions. This guide ranks the top 10 QMS platforms for medical device companies in 2026, evaluated on regulatory coverage, scalability, traceability, and fit-for-purpose design.
TL;DR
- A QMS platform digitizes the quality processes medical device companies must maintain to meet ISO 13485, FDA, and EU MDR requirements
- The right platform depends on company size, regulatory markets, and whether you need medical-device-specific workflows or a broader life sciences solution
- Key evaluation criteria: regulatory coverage natively supported, design control support, traceability, CAPA management, and total cost of ownership
- The top 10 platforms range from medtech-exclusive tools like Greenlight Guru to configurable enterprise systems like MasterControl and Veeva
- Selecting a platform is only half the work — implementation, SOP alignment, and gap analysis against ISO 13485 or FDA requirements typically determine whether you pass your first audit
What Is a QMS Platform and Why Do Medical Device Companies Need One?
A QMS platform is the structured digital framework that governs how a medical device company documents, controls, and demonstrates compliance with regulatory requirements across the product lifecycle. Unlike general manufacturing QMS tools, medical device platforms are tied directly to patient safety, regulatory clearance (510(k), PMA, CE marking), and post-market surveillance obligations.
Getting there is neither fast nor cheap. For small and mid-sized manufacturers, the path to a compliant QMS typically looks like this:
- Timeline: ISO 13485 certification spans 6–18 months depending on company maturity, with at least 2–3 months of operating records required before Stage 2 audits
- Cost: One-time certification runs $22,000–$190,000 when factoring in implementation, auditing, and certification body fees
The platforms reviewed below are assessed on regulatory coverage, validation support, scalability, and real-world fit. Each entry notes which company stage it suits best — whether you're a startup standing up your first eQMS or a global manufacturer coordinating compliance across FDA, EU MDR, and MDSAP jurisdictions.
Must-Have Features in a Medical Device QMS Platform
Choosing the wrong QMS platform can stall regulatory submissions and create audit vulnerabilities. Before evaluating vendors, confirm each platform covers these core functional modules:
- Document control with 21 CFR Part 11-compliant electronic signatures
- Design controls covering Design History Files (DHF), Device Master Records (DMR), and Device History Records (DHR)
- CAPA and nonconformance management to investigate and resolve quality events
- Risk management aligned with ISO 14971:2019, integrating risk analysis across the product lifecycle
- Training management to track personnel qualification and competency records
- Audit management for internal audits and third-party audit preparation
- Supplier quality management to control outsourced processes and components
- Post-market surveillance support for complaint handling, vigilance reporting, and trend analysis

Generic quality tools can check most of those boxes — but two capabilities set purpose-built medical device platforms apart:
- Native traceability linking user needs, design inputs, design outputs, verification/validation records, and risk analyses to quality events in closed-loop workflows
- Pre-validated or validation-ready infrastructure that reduces Computer System Validation (CSV) burden by providing IQ/OQ/PQ protocols and audit trail functionality
Cloud-based SaaS platforms now dominate this space — and for good reason. Faster deployment, automatic regulatory updates, and built-in remote audit readiness make them practical for companies managing compliance across FDA, EU MDR, Health Canada, TGA, and MDSAP jurisdictions simultaneously.
Top 10 QMS Platforms for Medical Device Companies in 2026
Each platform below is evaluated on regulatory coverage, design control and traceability capabilities, ease of implementation, and fit across company sizes—from first-time eQMS adopters to global multi-site manufacturers. Use this breakdown to shortlist platforms aligned to your stage, team size, and compliance requirements.
Greenlight Guru
Founded in 2013 and purpose-built exclusively for medical device companies, Greenlight Guru supports the full device lifecycle with pre-configured workflows for ISO 13485, FDA 21 CFR Part 820/QMSR, ISO 14971, and EU MDR compliance.
Why it stands out: Closed-loop traceability across design controls, risk management, and quality events is a genuine differentiator, and audit-tested templates meaningfully reduce time to first compliance milestone. That said, limited configurability and mandatory multi-year contracts make it a harder fit once companies move past the startup phase.
| Attribute | Details |
|---|---|
| Key Features | Document control, design controls, risk management, CAPA, complaint handling, audit management, post-market surveillance |
| Best For | Early-stage and growth-stage medical device startups establishing their first eQMS |
| Pricing | Pricing is not publicly disclosed; user reviews suggest premium pricing with mandatory multi-year commitments. Contact vendor for quote. |
MasterControl
Established in 1993 and headquartered in Salt Lake City, Utah, MasterControl is one of the most widely adopted QMS platforms in life sciences, serving over 1,000 clients globally including pharmaceutical, biotech, and medical device organizations.
Why it stands out: Tightly connected modules across document management, training, CAPA, and audit management give enterprise teams a unified compliance view. Validation on Demand via MasterControl's GxP platform reduces CSV burden. Complex implementation timelines and a steep learning curve, however, make it better suited for large organizations than SMEs.
| Attribute | Details |
|---|---|
| Key Features | Document control, training management, CAPA, complaint management, audit management, AI-driven workflows, batch approvals |
| Best For | Large medical device and pharma enterprises with complex, multi-site compliance requirements |
| Pricing | Enterprise pricing; typically $40,000+/year plus additional implementation and consultancy fees. Contact vendor for detailed quote. |
Qualio
Founded in 2012 and cloud-based, Qualio is designed for small- to mid-sized life science companies navigating FDA, ISO 13485, and GxP requirements, with a focus on ease of use and fast implementation.
Why it stands out: Intuitive UI, pre-built quality and regulatory content, AI compliance gap analysis (Compliance Intelligence), and integration with tools like Jira and Salesforce. Note limitations—documents must be recreated in Qualio's native format, and costs escalate with additional features and onboarding.
| Attribute | Details |
|---|---|
| Key Features | Document control, CAPA, training management, supplier management, compliance management, AI gap analysis, e-signatures |
| Best For | Venture-backed and fast-growing life science SMEs needing quick setup and structured templates |
| Pricing | Pricing is not publicly disclosed. Contact vendor for quote. |
QT9 QMS
QT9 QMS is a purpose-built, fully validated, cloud-based QMS platform for regulated manufacturers, with a dedicated medical device module offering out-of-the-box alignment with ISO 13485, FDA 21 CFR Part 820/QMSR, and EU MDR.
Where it excels: Comes pre-validated (reducing CSV burden), integrates with an optional ERP module for DHF/DMR/DHR and production traceability, and is frequently cited for ease of use and responsive customer support. A strong fit for companies that want a single validated vendor covering quality, operations, and compliance—without the overhead of a full enterprise deployment.
| Attribute | Details |
|---|---|
| Key Features | Document control, CAPA, design controls, risk management, training management, audit management, supplier quality, ERP integration, pre-validated CSV |
| Best For | Small to large medical device manufacturers needing a validated, integrated QMS with optional ERP connectivity |
| Pricing | Modular licensing model; mid-market pricing. Free trial available. Contact vendor for detailed quote. |
Veeva Vault Quality
Veeva Vault Quality is a cloud-native QMS platform built on the Vault architecture, widely adopted by large pharmaceutical and biotech organizations and designed for multi-site global compliance across GMP, GxP, and life sciences regulations.
Why it stands out: Deep native integration across Vault's regulatory, clinical, safety, and quality applications is the platform's core strength. AI agents launching across all Vault applications in April 2026 add further automation potential. The trade-off: it is optimized primarily for pharma/biotech workflows, implementation is typically multi-year and consultant-dependent, and organizations outside the Veeva ecosystem face steep costs to unlock full value.
| Attribute | Details |
|---|---|
| Key Features | Document management, CAPA, audit management, training, complaint handling, regulatory submissions integration, AI agents, GxP compliance |
| Best For | Large pharma and biotech enterprises already using Veeva Vault for regulatory or clinical operations |
| Pricing | Enterprise-tier; typically six-figure annual investments. Contact vendor for quote. |
TrackWise Digital (Sparta Systems / Honeywell)
With over 30 years of life sciences QMS heritage, TrackWise Digital is an enterprise-grade platform enhanced with AI-augmented workflows and Honeywell manufacturing integration, widely used across global pharma and biotech organizations.
Why it stands out: Strong multi-site harmonization, AI-assisted event auto-categorization, CAPA and complaint analytics. However, configuration for modules like document management and training may require vendor support, and UI reviews are mixed among users—better suited for mature quality organizations than companies establishing their first QMS.
| Attribute | Details |
|---|---|
| Key Features | Document management, CAPA, audit management, deviation management, complaints, PMS, AI-assisted analytics, multi-site quality harmonization |
| Best For | Global pharma and biotech enterprises with mature quality organizations and complex multi-site operations |
| Pricing | Enterprise-level; typically requires vendor quote. |
ETQ Reliance
ETQ Reliance (now branded as Octave Reliance under Hexagon AB) is a highly configurable, cloud-based QMS platform widely used in life sciences, pharmaceuticals, and medical device manufacturing, known for its flexibility and strong support for complex global deployments.
Key differentiator: Comprehensive coverage across nonconformance, CAPA, supplier quality, complaints, eMDR, and validation, with specialized 21 CFR Part 11 compliance apps. That configurability is a real asset—but it requires mature quality organizations with dedicated IT resources to leverage it well. Teams seeking out-of-the-box simplicity should look elsewhere.
| Attribute | Details |
|---|---|
| Key Features | Nonconformance management, CAPA, supplier quality, audit management, complaints, eMDR, 21 CFR Part 11 compliance, change control |
| Best For | Mid-to-large medical device and life sciences companies with complex supply chains needing high configurability |
| Pricing | Quote-based pricing. Contact vendor for details. |
ComplianceQuest
ComplianceQuest is a QMS platform built natively on Salesforce, targeting life sciences and manufacturing organizations—including medical device companies—that want a cloud-first, flexible, and integrated quality management solution.
Why it stands out: End-to-end quality workflow coverage (documents, training, NC/CAPA, complaints, audits) aligned to life sciences regulations; EHS module available for safety-intensive environments; AI-driven features for predictive risk and workflow automation. Note that pricing transparency has been flagged as a concern by some prospects.
| Attribute | Details |
|---|---|
| Key Features | Document control, CAPA, training, audit management, supplier quality, complaints, EHS module, AI-driven risk analytics, Salesforce-native architecture |
| Best For | Mid-to-large medical device and life sciences companies already using Salesforce or prioritizing EHS alongside quality |
| Pricing | Non-transparent; requires vendor engagement for quote. |
Cognidox
Founded in 2008 in Cambridge, UK, Cognidox is a lean eQMS platform designed for SMEs and innovation-driven companies in the medtech and high-tech sectors, with a document-centric approach to compliance and a lower cost of entry compared to enterprise alternatives.
Worth knowing: Flexible and affordable starting costs; full suite of QMS modules (CAPAs, training, supplier management, e-signatures) included as standard; built-in learning management system developed with medical device developers. However, automated traceability matrices must be built manually, and out-of-the-box integrations are limited to Microsoft Office and DocuSign.
| Attribute | Details |
|---|---|
| Key Features | Document control, CAPAs, training/LMS, supplier management, change management, e-signatures, design controls, software validation support |
| Best For | SMEs and scale-ups transitioning from paper or Google Docs to a flexible, right-sized eQMS |
| Pricing | Starts from approximately $9,000/year for 20 users. Contact vendor for current pricing. |
Q-Pulse (Ideagen)
Q-Pulse, now branded as Ideagen Quality Management, is a modular QMS platform headquartered in Nottingham, UK, serving regulated industries including life sciences, healthcare, aviation, and manufacturing with a comprehensive suite of quality and compliance tools.
Why it stands out: Versatile and configurable modular design supporting document control, CAPA, audits, risk management, and real-time product information management. However, its interface is considered less modern compared to newer platforms, and the steep learning curve and rising per-seat costs can be a barrier for smaller teams.
| Attribute | Details |
|---|---|
| Key Features | Document control, CAPA, audit management, risk management, training management, supplier quality, BOM tracking, product information management |
| Best For | Regulated companies across multiple industries needing a modular, configurable QMS with internal IT support |
| Pricing | Starts from approximately $20,000/year, rising with additional modules and seats. Contact vendor for current pricing. |
How to Choose the Right QMS Platform for Your Company
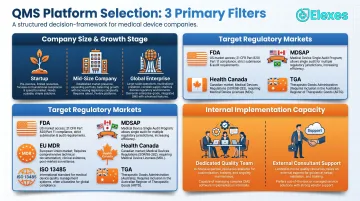
The most common mistake companies make is prioritizing brand reputation or feature count over fit—choosing an enterprise platform that's too complex to implement quickly, or a startup tool they'll outgrow within 18 months. Frame your evaluation around three primary filters:
- Company size and growth stage — Are you an early-stage startup establishing your first QMS, a mid-sized company scaling across multiple markets, or a global enterprise harmonizing multi-site operations?
- Target regulatory markets — FDA, EU MDR, ISO 13485, MDSAP, Health Canada, TGA?
- Internal implementation capacity — Do you have a dedicated quality team, or will you need external validation and ongoing compliance support?
Key Evaluation Criteria Tied to Business Outcomes
- Out-of-the-box regulatory alignment vs. configuration effort — Does the platform support your device type (SaMD, IVD, implantables) with pre-configured design control, DHF/DMR/DHR structures, risk management per ISO 14971, and post-market surveillance workflows?
- Total cost of ownership — Include implementation, training, validation, and per-seat scaling costs; one medtech consultancy estimates one-time QMS setup costs range from €50,000 to €150,000, with ongoing annual costs of €50,000 to €200,000 for mid-sized companies
- Audit readiness and validation documentation — Does the platform provide IQ/OQ/PQ templates, 21 CFR Part 11 compliance controls, and audit trails that hold up under FDA or Notified Body review?
Software selection is only part of the equation. Successful QMS implementation requires knowledgeable quality resources — internal or external. Companies without a dedicated quality team typically engage regulatory consultants to configure, validate, and maintain their platform against ISO 13485 and applicable regulations. Elexes provides exactly this kind of hands-on QMS support, helping clients across FDA, EU MDR, and MDSAP frameworks get their systems audit-ready from day one.
Conclusion
No single QMS platform is the right fit for every medical device company. The best choice aligns with your regulatory roadmap, company size, device type, and operational capacity—not just market visibility or peer recommendations.
Run demos and trials, stress-test shortlisted platforms against your actual quality workflows, and validate your selection against the regulatory markets you intend to serve. Bringing a QMS to audit-ready status takes meaningful time and resources — factor that in before committing.
For medical device companies that need guidance beyond software selection, Elexes provides QMS consulting services across gap assessments, implementation support, audit preparation, and post-market surveillance documentation. With 50+ years of collective experience and a 90% audit clearance rate across FDA, ISO 13485, EU MDR, and global frameworks, the team has supported 250+ projects for companies at every stage.
Frequently Asked Questions
What is the best QMS software for medical device companies?
There is no single "best" platform. Greenlight Guru suits medtech startups, MasterControl and Veeva suit large enterprises, and platforms like QT9 QMS and Qualio work well for mid-sized companies. The best choice depends on company size, regulatory markets, and internal implementation capacity.
What is QMS in MedTech?
A Quality Management System in MedTech is the structured set of processes, documents, and controls a medical device company maintains to meet regulations like ISO 13485, FDA 21 CFR Part 820/QMSR, and EU MDR. Core areas include design controls, CAPA, risk management, supplier quality, and post-market surveillance.
What are the two main quality systems for medical devices?
ISO 13485 (the international QMS standard for medical devices, widely required for CE marking and global market access) and FDA 21 CFR Part 820/QMSR (the U.S. regulatory requirement for device manufacturers). The FDA's 2024 QMSR update harmonized 21 CFR Part 820 with ISO 13485.
What are some QMS tools?
Common QMS tools used in the medical device industry include dedicated eQMS platforms (Greenlight Guru, MasterControl, QT9 QMS, Qualio, Veeva), as well as document control, CAPA management, risk management (ISO 14971-aligned), training management, and audit management modules—either standalone or bundled within an eQMS platform.
What are the 7 steps of QMS?
The general QMS implementation steps are:
- Define quality policy and objectives
- Conduct a gap analysis against the applicable standard, such as ISO 13485
- Document procedures and SOPs
- Implement and train staff
- Conduct internal audits
- Perform management review
- Pursue certification or regulatory approval
This is an iterative, ongoing process — not a one-time project.


