
Introduction
Poor document control ranks among the most common root causes cited in FDA warning letters and audit non-conformances across the medical device industry. According to FDA inspection data from FY2023, seven of the top 10 most-cited observations were documentation-related, with CAPA documentation failures alone appearing 398 times. These failures aren't just administrative oversights—they represent fundamental quality system breakdowns that can delay product launches, trigger costly remediation, and directly threaten patient safety.
Document control consultants help medical device companies build compliant systems for Standard Operating Procedures (SOPs), Design History Files (DHF), change control processes, and version management. All are required for ISO 13485 and FDA Quality Management System Regulation (QMSR) compliance. With the FDA QMSR now in full effect as of February 2, 2026, manufacturers must demonstrate compliance with ISO 13485:2016 document control clauses during inspections.
Non-routine quality events cost the US medical device industry between $7.5 billion and $9 billion annually, with individual manufacturers spending more than $250,000 on remediation for a single set of observations. Getting document control right before an inspection costs significantly less than fixing it after one.
The firms below were selected based on audit success rates, depth of regulatory expertise, and demonstrated QMS support across device classes and global markets — to help you find the right partner before your next inspection.
TL;DR
- Document control consulting covers SOPs, DHF/DMR/DHR records, change control, and version management under ISO 13485 and FDA QMSR
- Critical hiring windows: QMS build-out, pre-audit prep, regulatory submissions, and post-market surveillance
- Elexes tops this list — 90%+ audit clearance rate, full QMS lifecycle support across FDA, EU MDR, and global standards
- Rankings weighted by document control depth, ISO 13485/QMSR alignment, audit track record, and engagement flexibility
- Your best fit depends on device class, target markets, and where your documentation stands today
What Is Document Control Consulting for Medical Device Companies?
Document control in the medical device context covers the full lifecycle of controlled documents: creation, review, approval, versioning, distribution, and retirement. This includes SOPs, work instructions, DHFs, Device Master Records (DMR), Device History Records (DHR), risk files, and CAPAs.
The system must satisfy ISO 13485:2016 Section 4.2, FDA 21 CFR Part 820.40, and the updated QMSR requirements now in effect.
Common pain points that drive companies to outsource document control:
- Inadequate SOP structure lacking clear approval workflows
- Uncontrolled document versions circulating across multiple departments
- Missing electronic signature trails required under 21 CFR Part 11
- Poor change control workflows that fail to track document modifications
- Audit-readiness gaps discovered only during inspections
These issues hit hardest at three stages: startups building their first QMS, scaling companies entering new regulatory markets, and established manufacturers remediating post-inspection findings. It's a widespread problem — document control ranks as the #1 most common non-conformity in MDSAP audits across all five participating jurisdictions.

The consultants listed below were selected based on document control-specific capabilities, evaluated on technical depth rather than general regulatory brand recognition.
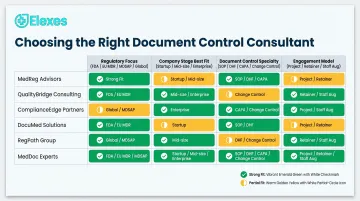
Top 10 Document Control Consultants for Medical Device Companies in 2026
The following consultants were selected based on document control-specific expertise, ISO 13485/QMSR/EU MDR alignment, audit clearance track records, engagement flexibility, and client base credibility—not brand size alone.
1. Elexes
Elexes is a regulatory and quality consulting firm with a 50+ years of collective expertise, having supported 256+ medical device clients across the US, UK, EU, Asia, and beyond. The firm helps companies build audit-ready QMS systems and controlled document environments—covering SOP creation, DHF/DMR management, CAPA documentation, and change control support.
| Key Services | SOP creation, DHF/DMR/DHR documentation, CAPA management, change control support, QMS gap analysis, compliance audits, ISO 13485/MDSAP/FDA QMSR alignment |
| Compliance Expertise | ISO 13485, ISO 14971, IEC 62304, FDA 21 CFR Part 820/QMSR, EU MDR, MDSAP, MHRA, Health Canada |
| Best For | Startups to global device manufacturers needing flexible, project-based or ongoing document control and QMS support across multiple regulatory jurisdictions |

2. Cannon Quality Group
Founded in 2010, Cannon Quality Group is a full-service outsourced quality management firm specializing in document-controlled QMS environments for MedTech, IVDR, and SaMD companies. Their track record includes supporting 200+ companies with 25 employees and a notable Audit Efficiency Program.
Their Audit Efficiency Program runs shared supplier audits that generate reusable documentation across 15-20 clients simultaneously, reducing per-client audit costs significantly. The firm focuses on replacing reactive, fear-based quality systems with practical, maintainable document controls built for long-term compliance.
| Key Services | QMS build-out, document control program setup, supplier audit documentation, ISO 13485 compliance, change control workflows |
| Compliance Expertise | ISO 13485, FDA 21 CFR Part 820, IVDR, SaMD documentation requirements |
| Best For | MedTech startups and mid-size companies needing outsourced QMS document management with a lean, practical approach |
3. QES Medical
Boston-based QES Medical brings 17+ years of experience supporting medical device and pharmaceutical companies with QMS building, regulatory submissions, and design control documentation. Led by industry veteran Oscar Garcia, the firm draws on leadership experience from multiple Boston-area device companies.
QES Medical is particularly strong in FDA-facing document control, including 21 CFR Part 820/QMSR-compliant SOPs, Design History File structuring, and audit readiness documentation. Their risk management documentation support (ISO 14971) adds depth for companies handling complex device filings and design control requirements.
| Key Services | QMS building, SOP development, FDA compliance documentation, design and development records, risk management files, audit readiness |
| Compliance Expertise | ISO 13485, FDA 21 CFR Part 820/QMSR, ISO 14971, SaMD documentation |
| Best For | US-based companies and startups needing hands-on FDA-facing document control and QMS support |
4. Rook Quality Systems
With over 12 years of experience, Rook Quality Systems offers certified quality auditor-led document control and QMS support for hardware, SaMD, and IVD medical device companies ranging from Class I to Class III.
Their team of 13+ certified quality auditors brings deep ISO 13485 and MDSAP documentation expertise, making them particularly valuable for companies preparing for multi-jurisdictional audits where document traceability and record integrity are under scrutiny. Their auditor perspective helps manufacturers anticipate and address documentation gaps before inspections.
| Key Services | ISO 13485 document control, MDSAP documentation, EU MDR CE marking records, QMS audit preparation |
| Compliance Expertise | ISO 13485, MDSAP, EU MDR, FDA 21 CFR Part 820 |
| Best For | Companies undergoing MDSAP or ISO 13485 audits who need certified auditor-reviewed document systems |
5. MWA Consulting / AVS Life Sciences
MWA Consulting, now part of AVS Life Sciences following a January 2024 acquisition, operates with 300+ associates across the US, Canada, India, South America, and Europe, offering QMS-building and document control services to life science companies navigating GxP environments.
Their breadth of international associates makes them valuable for companies managing controlled document systems across multiple regulatory zones. Their risk management documentation capabilities (FMECA, FTA, hazard analysis) complement core document control programs, particularly for manufacturers with distributed operations.
| Key Services | QMS documentation, preventive maintenance records, risk management files, internal audit documentation, 510(k) submission support |
| Compliance Expertise | GxP, ISO 13485, FDA 21 CFR Part 820, risk management standards |
| Best For | Multi-site or global device companies needing distributed document control support across regulatory regions |
6. Emergo by UL
Emergo by UL operates across 25+ countries with comprehensive document control and QMS compliance services backed by proprietary tools like RAMS® and OPUS™.
Their scale enables them to support complex, multi-market document control programs—particularly for companies seeking simultaneous compliance with FDA, EU MDR, and Asia-Pacific regulatory frameworks. Their global presence and digital tools make them particularly strong for large enterprises managing high document volumes across international operations.
| Key Services | Global QMS implementation, document control program design, regulatory strategy, device registration documentation |
| Compliance Expertise | FDA, EU MDR, ISO 13485, IVDR, TGA, Health Canada, PMDA |
| Best For | Large or global medical device manufacturers entering multiple markets simultaneously |
7. Regulatory Compliance Associates (RCA)
Regulatory Compliance Associates is composed largely of former FDA officials and industry veterans, giving their document control and QMS remediation services a uniquely regulatory-insider perspective. This expertise is particularly valuable when companies face warning letters, 483 observations, or pre-submission document gaps.
RCA specializes in mock audits, document remediation, and CAPA documentation, helping companies bring non-conforming document systems back into full compliance with FDA and ISO standards. That insider view of how FDA reviewers interpret documentation translates directly into faster, more targeted remediation.
| Key Services | Document remediation, mock audit preparation, CAPA documentation, QMS gap analysis, SOP review |
| Compliance Expertise | FDA 21 CFR Part 820/QMSR, ISO 13485, QSR compliance |
| Best For | Companies responding to FDA observations or warning letters who need expert document remediation support |
8. Effectum Medical
Switzerland-based Effectum Medical offers a "plug-and-play" eQMS document package: pre-built SOPs and templates delivered within a digital QMS platform. This approach makes rapid document control implementation accessible for early-stage companies entering the EU market.
Their ability to act as both a document control consultant and legal manufacturer/Authorized Representative for EU access gives them a dual-role advantage. Their pre-validated document library accelerates time-to-compliance for startups with limited internal resources who need structured documentation quickly.
| Key Services | Pre-built SOP library, eQMS document set, EU MDR/IVDR document control, Authorized Representative services |
| Compliance Expertise | ISO 13485, EU MDR, IVDR, FDA |
| Best For | Startups and early-stage companies needing fast, structured document control infrastructure for EU market entry |
9. Medpoint
Medpoint is a US-based consultancy with a global team of 350 consultants, offering QA and document control support across the full device lifecycle—from initial design documentation to post-market surveillance records and global supplier audit files.
Their breadth of consultants and geographic coverage makes them well-suited for scaling medical device companies that need ongoing document control staffing augmentation without the cost of full-time headcount. Their flexible staffing model allows companies to scale document control resources up or down based on project needs.
| Key Services | QA documentation, FDA 21 CFR 820/QMSR records, MDSAP compliance documentation, clinical document support, supplier audit records |
| Compliance Expertise | FDA, ISO 13485, MDSAP, global regulatory standards |
| Best For | Rapidly scaling device companies needing flexible document control staffing across multiple regulatory regions |
10. NorMed Consulting
Copenhagen-based NorMed Consulting offers remote and hybrid document control and regulatory compliance support for medical device companies targeting the EU, UK, and US markets—with particular strength in SaMD, AI-driven device documentation, and combination product records.
Their right-first-time documentation philosophy—building document structures that are simultaneously compliant with EU MDR, UKCA, and FDA QMSR—is especially valuable for companies managing tri-market submissions with a lean internal team. Their SaMD specialization addresses the unique documentation challenges of software-driven devices.
| Key Services | Document control system setup, SaMD documentation, EU MDR/UKCA/FDA multi-market records, risk and clinical documentation |
| Compliance Expertise | EU MDR, UKCA, FDA QMSR, ISO 13485, IEC 62304 (SaMD) |
| Best For | SaMD and combination device companies targeting EU, UK, and US markets simultaneously |
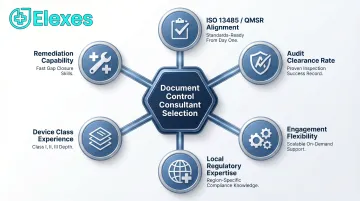
How We Chose the Best Document Control Consultants
We assessed consultants based on document control-specific capabilities—not just general regulatory reputation. The most common mistake companies make is choosing consultants for brand recognition rather than validating their specific document control track record and audit success rates.
Key selection criteria included:
- Demonstrated procedures meeting ISO 13485:2016 Clauses 4.2.4 (document control) and 4.2.5 (record control), aligned with FDA QMSR/21 CFR Part 820
- Verified audit clearance rates across ISO 13485, MDSAP, FDA, and EU MDR inspections without major non-conformances
- Flexible engagement models — project-based, retainer, and staff augmentation — to match budget and scope
- Local expertise and language capabilities across target regulatory markets
- Proven track record across device classes (I, II, III), product types (hardware, SaMD, IVD, implantables), and company stages
- Capacity to support both early-stage QMS build-outs and large-scale document remediation following FDA observations
Based on FDA inspection data, approximately 75-80% of the most frequently cited observations use language like "procedures not established," "not documented," or "not maintained." The primary value of document control consulting is establishing and documenting those procedures before an inspection occurs.
Three factors determine the best consultant for your company:
- Product lifecycle stage: Early-stage QMS build-out requires different support than post-market remediation
- Target regulatory markets: FDA-only submissions need different documentation than tri-market (US/EU/UK) strategies
- Engagement scope: Full QMS outsourcing versus targeted document control gap remediation
Conclusion
Document control is not a back-office function—it is the foundational evidence layer connecting regulatory compliance to patient safety. Choosing a consultant who treats it strategically—rather than as a paperwork exercise—determines whether your QMS holds up when regulators arrive.
When evaluating candidates, focus on practical deliverables rather than credentials alone:
- Can they show you sample SOP structures aligned to your device class?
- Have they supported companies in your specific risk category and target markets?
- Do they understand 2026 regulatory changes like FDA QMSR enforcement and evolving EU MDR documentation requirements?
With FDA QMSR enforcement now active and document control consistently cited as a top audit non-conformity globally, the cost of getting this wrong is higher than ever.
Elexes provides document control and QMS consulting for medical device companies across US, EU, and global markets. With 50+ years of collective team experience, a 90% audit clearance rate, and 250+ completed projects, they support everything from SOP development to full QMS implementation. Contact Elexes to discuss your document control program.
Frequently Asked Questions
What is the best way to create SOPs for medical device document control?
Medical device SOPs should follow a controlled template aligned to ISO 13485:2016 Section 4.2 and FDA 21 CFR Part 820.40, covering purpose, scope, responsibilities, procedures, and approval signatures. Each document must be version-controlled, linked to the correct QMS tier, and reviewed whenever processes or regulations change.
What does a document control consultant do for a medical device company?
Document control consultants design and implement document control systems including SOPs, DHF, DMR, DHR, and change control workflows. They train staff on Good Documentation Practices (GDocP), prepare controlled document environments for ISO 13485 or FDA QMSR audits, and remediate non-conforming documentation identified during inspections.
What regulations require document control for medical devices?
The primary requirements come from:
- ISO 13485:2016 Section 4.2 — documents and records control
- FDA 21 CFR Part 820.40 / QMSR — document approval and change control
- FDA 21 CFR Part 11 — e-signatures in electronic records
- EU MDR Annex IX — technical documentation
Document control deficiencies consistently rank among the most cited audit observations worldwide.
What is the difference between document management and document control for medical devices?
Document management covers general file storage, sharing, and retrieval. Document control is the regulated process of approving, versioning, distributing, and retiring documents through defined workflows — mandated by ISO 13485 and FDA QMSR, with formal audit trails and e-signature requirements.
When should a medical device company hire a document control consultant?
The most critical trigger points are during initial QMS setup before first regulatory submission, when preparing for an ISO 13485 or FDA audit, when entering a new regulatory market with different documentation requirements, or after receiving audit observations citing documentation deficiencies.
How much does document control consulting for medical devices typically cost?
Pricing depends on scope: SOP development and gap assessments are typically fixed-fee, while ongoing QMS support runs as monthly retainers or hourly contracts. Request a proposal tied to specific deliverables rather than comparing hourly rates alone for accurate budget planning.


