
Introduction
Adverse event reporting failures rank among the leading triggers of FDA warning letters and EU MDR non-conformities for medical device companies. A 2025 FDA warning letter to Royal Philips cited failures to submit malfunction MDRs under 21 CFR 803.50 and systemic complaint-handling deficiencies, illustrating how a single missed report can spiral into major regulatory enforcement.
A BMJ cross-sectional study of 4.4 million manufacturer reports found that 29.1% were not demonstrably submitted on time, with just three manufacturers accounting for 54.8% of late reports.
With FDA 21 CFR Part 803, EU MDR Article 87, MDSAP, TGA IRIS, and MHRA vigilance requirements all tightening, companies without dedicated AER expertise risk product delays, recalls, and market access loss. This guide covers the top 10 adverse event reporting consultants for 2026 — what each does well, who they serve best, and how to choose the right fit for your situation.
TL;DR
- Adverse event reporting is mandatory under FDA, EU MDR, MHRA, Health Canada, and TGA—non-compliance triggers warning letters, consent decrees, or market removal
- Specialized AER consultants help identify reportable events, meet jurisdiction-specific timelines, and build sustainable post-market surveillance programs
- The right consultant brings cross-regulatory expertise, hands-on MDR submission experience, and engagement models that scale with your team's needs
- Each firm on this list was evaluated for AER depth, global regulatory coverage, and track record with complaint handling and PMS systems
What Is Adverse Event Reporting for Medical Devices?
Adverse event reporting (AER) is the regulatory requirement to identify, document, and submit reports to authorities when a device may have caused or contributed to patient harm, death, or serious malfunction. Under FDA 21 CFR Part 803, manufacturers must report deaths or serious injuries within 30 calendar days — or 5 working days when remedial action is needed to prevent substantial harm.
EU MDR Article 87 sets tighter deadlines: 2 calendar days for public health threats, 10 days for death or serious deterioration, and 15 days for other serious incidents.
Key global frameworks include:
- FDA MDR (US): 30-day standard reporting; 5-day urgent reports for imminent harm
- EU MDR Article 87: 2/10/15-day timelines submitted via EUDAMED
- MHRA MORE system (UK): 2/10/15-day timelines, mirroring EU MDR post-Brexit
- Health Canada Vigilance: 10 days for death/serious deterioration; 30 days for potential serious harm
- TGA IRIS (Australia): 48 hours for serious public health threats; 10 days for death/serious deterioration

Keeping pace with these overlapping timelines is where many internal teams fall short. Jurisdiction-specific expertise is hard to maintain in-house, reporting volume spikes unpredictably post-launch, and a single missed or late submission can trigger regulatory scrutiny. For most manufacturers, outsourcing AER is a risk management decision.
Top 10 Adverse Event Reporting Consultants for Medical Device Companies in 2026
These firms were evaluated on regulatory coverage, AER-specific capabilities, certifications, client track record, and ability to support both startup and established manufacturers. Use the breakdowns below to identify which firm aligns with your reporting obligations, device category, and target markets.
Elexes
Background: Elexes is a global medical device regulatory consulting firm with 50+ years of collective team experience, supporting 100+ clients across the US, EU, UK, Canada, and Asia-Pacific with full-cycle regulatory and post-market compliance services including adverse event reporting and post-market surveillance.
Standout factor: Elexes offers cross-functional outsourcing that combines regulatory, quality, and PMS tasks under one engagement—including adverse event identification, MDR submission support, FSCA planning, and complaint handling aligned to FDA 21 CFR Part 803, EU MDR, MDSAP, and MHRA requirements. With 250+ successful projects and a 90% audit clearance rate, they bring measurable outcomes without adding permanent headcount.
| AER Capabilities | Coverage & Credentials |
|---|---|
| MDR submissions, FSCA support, complaint handling, PMS reporting, CAPA documentation, vigilance reporting across FDA, EU MDR, MHRA, and Health Canada | USA, EU, UK, Canada, Australia, Saudi Arabia, Asia-Pacific |
| ISO 13485, MDSAP, GCP, HIPAA certified | Flexible engagement models: full-time, part-time, or project-based; complete data confidentiality |

USDM Life Sciences
Background: USDM Life Sciences is a US-based life sciences consulting firm with 20+ years of experience, offering dedicated complaint handling and adverse event management outsourcing to biotech, pharma, and medical device companies.
Standout factor: USDM provides a structured outsourced team model—assigning post-market surveillance specialists, investigators, and clinicians to augment in-house teams, with assigned team leads, daily progress reviews, and weekly performance metrics. Their work on a 600,000+ service order backlog remediation for a global device manufacturer demonstrates large-scale AER delivery.
| Category | Details |
|---|---|
| Key AER Services | Complaint coding, AE investigation, CAPA initiation, MDR reporting, EU MDR recoding, consent decree support |
| Regulatory Markets Covered | USA, EU (MDR/IVDR) |
| Notable Certifications / Differentiators | GxP-exclusive focus; managed services delivery model; large team scalability |
Celegence
Background: Celegence is a global regulatory consulting firm specializing in medical devices and pharmaceuticals, with dedicated service lines for post-market surveillance, PMCF, and vigilance reporting under EU MDR and FDA frameworks.
Standout factor: Celegence's AI-powered CAPTIS® platform streamlines adverse event database searches (including MAUDE and TPLC) and supports PSUR and PMSR authoring. Integration with systematic literature reviews reduces PMS documentation turnaround time. Their EU MDR technical team brings specialized CER and clinical evaluation authoring depth.
| Category | Details |
|---|---|
| Key AER Services | PMS reporting, PSUR, PMSR, PMCF, vigilance/MDR authoring, complaint management |
| Regulatory Markets Covered | EU (MDR/IVDR), USA (FDA), UK (MHRA) |
| Notable Certifications / Differentiators | Proprietary CAPTIS® AI platform; specialized EU MDR expertise; CER and SLR integration with PMS |
QES Medical
Background: Boston-based QES Medical, founded in 2009, provides quality, regulatory, and SaMD consulting services to medical device and pharmaceutical companies, with capabilities in FDA compliance and QMS-based adverse event handling.
Standout factor: QES Medical's Health Hazard Evaluation (HHE) expertise directly supports AER triage—helping manufacturers determine whether a device malfunction crosses the MDR reportability threshold under 21 CFR Part 803, a step where many manufacturers face compliance gaps.
| Category | Details |
|---|---|
| Key AER Services | Health Hazard Evaluations, FDA MDR compliance, QMS-integrated AE handling, risk management, SaMD vigilance |
| Regulatory Markets Covered | USA (FDA), ISO 13485 globally |
| Notable Certifications / Differentiators | ISO 13485, FDA 21 CFR 820 expertise; SaMD and IVD experience; 20+ years of FDA regulatory leadership |
Rook Quality Systems (RookQS)
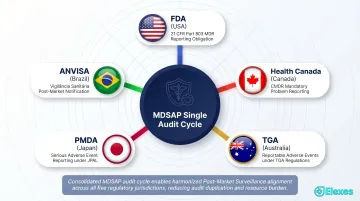
Background: RookQS is a quality-focused medical device consultancy founded in the early 2010s, with a team of experts including 13 certified quality auditors, serving Class I–III device manufacturers across hardware, SaMD, and IVD categories.
Standout factor: RookQS has deep MDSAP audit expertise, which integrates adverse event and complaint handling requirements from five regulatory jurisdictions (FDA, Health Canada, ANVISA, TGA, PMDA) into a single audit cycle—valuable for manufacturers seeking multi-market PMS alignment.
| Category | Details |
|---|---|
| Key AER Services | MDSAP-aligned complaint handling, QMS AE integration, EU MDR vigilance support, ISO 13485 QMS audits |
| Regulatory Markets Covered | USA, Canada, Australia, Japan, Brazil (MDSAP jurisdictions); EU MDR |
| Notable Certifications / Differentiators | 13 certified quality auditors; MDSAP specialization; hardware, SaMD, and IVD coverage |
Cannon Quality Group
Background: Cannon Quality Group, founded in 2010, is a full-service outsourced quality management firm for medtech, IVDR, and SaMD companies, known for its QMS-first approach to embedding compliant complaint and adverse event processes.
Standout factor: Cannon's proprietary Audit Efficiency Program—where multiple clients' supplier audits are conducted in a single session—extends to complaint handling infrastructure, helping companies build lean, scalable AER processes without duplicating compliance overhead.
| Category | Details |
|---|---|
| Key AER Services | QMS-integrated AE handling, complaint process design, CAPA management, outsourced quality management |
| Regulatory Markets Covered | USA (FDA), EU (MDR/IVDR), SaMD globally |
| Notable Certifications / Differentiators | Audit Efficiency Program; long-term outsourced QMS model; SaMD and IVDR-specific experience |
Medpoint
Background: US-headquartered Medpoint offers a broad-spectrum medical device consulting service with a global team of 350 consultants, covering QA, regulatory, clinical support, and supplier auditing for device manufacturers at all lifecycle stages.
Standout factor: Medpoint's team of 350 consultants and global supplier auditing network supports large manufacturers managing high-volume adverse event workloads across multiple geographies simultaneously, covering EU MDR vigilance and FDA 21 CFR 820 compliance.
| Category | Details |
|---|---|
| Key AER Services | FDA 21 CFR 820 QA support, MDSAP compliance, EU MDR vigilance, global supplier audits, clinical consulting |
| Regulatory Markets Covered | USA, EU, Canada, global |
| Notable Certifications / Differentiators | 350+ global consultants; full-service clinical and regulatory integration; suitable for large-portfolio manufacturers |
NorMed Consulting
Background: Copenhagen-based NorMed Consulting is a specialist medtech consultancy offering remote or hybrid engagement models for SaMD, AI-based devices, combination products, and classic hardware companies targeting EU, UK, and US markets.
Standout factor: NorMed's EU MDR and UK MHRA compliance work covers PMS plan design and vigilance reporting from first submission through recertification. Their remote-first model suits SaMD and AI device manufacturers navigating post-market obligations for European market entry without the overhead of a full in-country team.
| Category | Details |
|---|---|
| Key AER Services | EU MDR PMS planning, MHRA vigilance, SaMD and AI device post-market compliance, combination product AER |
| Regulatory Markets Covered | EU (MDR), UK (MHRA), USA (FDA) |
| Notable Certifications / Differentiators | SaMD and AI device specialization; remote-first flexible engagement; EU/UK market entry focus |
Platypus MedTech Consulting
Background: Australia-based Platypus MedTech Consulting is led by regulatory expert Dr. Lee Walsh and specializes in TGA submissions, ISO compliance, digital health, cybersecurity, and SaMD regulatory pathways for the Australian and international markets.
Standout factor: Platypus brings niche expertise in TGA's IRIS adverse event reporting scheme combined with cybersecurity and digital health regulatory knowledge—a differentiated offering for connected device and wearable manufacturers who face both PMS and cybersecurity vigilance obligations.
| Category | Details |
|---|---|
| Key AER Services | TGA IRIS adverse event reporting, ISO 13485 compliance, SaMD and digital health vigilance, cybersecurity regulatory support |
| Regulatory Markets Covered | Australia (TGA), ISO globally, SaMD international |
| Notable Certifications / Differentiators | TGA and ISO deep specialization; cybersecurity + AER integration; digital health and wearable device focus |
Emergo by UL
Background: Emergo by UL is a medical device regulatory consulting firm operating across 40+ countries, offering regulatory strategy, submissions, post-market surveillance, and vigilance services for device manufacturers entering or sustaining market access worldwide.
Standout factor: Emergo covers FDA, EU MDR, PMDA, NMPA, Health Canada, and CDSCO frameworks under one engagement. For manufacturers with multi-country adverse event reporting obligations, that consolidation eliminates the coordination overhead of managing separate regional consultants.
| Category | Details |
|---|---|
| Key AER Services | Global vigilance reporting, PMS plan authoring, MDR submissions, FSCA coordination, EU MDR Article 87 compliance |
| Regulatory Markets Covered | USA, EU, UK, Japan, China, India, Canada, Australia, 40+ countries |
| Notable Certifications / Differentiators | UL brand global infrastructure; 40+ country regulatory coverage; strong APAC and emerging market reach |
How We Chose the Best Adverse Event Reporting Consultants
Our assessment focused on demonstrated AER-specific capabilities—not just general regulatory consulting—including complaint investigation protocols, MDR/vigilance submission experience, and PMS system integration. The most common mistake companies make is selecting general QMS consultants who lack jurisdiction-specific AER expertise.
Key selection factors:
- Multi-jurisdictional regulatory coverage (FDA, EU MDR, MDSAP, MHRA, TGA)
- Flexibility of engagement model (project-based vs. long-term outsourcing)
- Certifications relevant to AER (ISO 13485, MDSAP, GCP)
- Evidence of successful audit clearance or remediation outcomes
The right fit depends on where you are in your regulatory journey. A startup launching its first 510(k) needs a firm with hands-on submission experience and scalable bandwidth. A large manufacturer managing EU MDR recertification across multiple markets needs deep vigilance expertise and proven backlog remediation. Matching firm specialization to your specific situation is what separates a productive engagement from a costly one.
Conclusion
Adverse event reporting is a mandatory post-market obligation under every major global regulatory framework. The consequences of underreporting or missed timelines—including FDA warning letters, EUDAMED flags, or market suspension—make expert consulting support a business-critical investment rather than optional overhead.
Assess your current AER workload, target markets, and internal capacity before selecting a consultant. Prioritize firms that offer both regulatory expertise and operational infrastructure—team models, reporting templates, escalation protocols—rather than just advisory guidance.
Elexes provides end-to-end adverse event reporting and post-market surveillance support across FDA, EU MDR, MDSAP, MHRA, and Health Canada frameworks, with flexible engagement models and a 90% audit clearance rate across 250+ projects. Reach out via +1 408-475-8091 or through the Elexes website to discuss your PMS and AER compliance needs.
Frequently Asked Questions
Who will report any adverse incidents relating to medical devices?
Manufacturers, importers, and authorized representatives hold the primary legal obligation to report adverse events to regulators under FDA 21 CFR Part 803 and EU MDR Article 87. Healthcare providers and patients may report voluntarily in most jurisdictions; in the EU, healthcare professionals are encouraged—but not universally mandated—to do so.
What is a vigilance medical device?
"Medical device vigilance" refers to the post-market system manufacturers maintain to detect, assess, and report serious incidents. It is the EU/international equivalent of the FDA's Medical Device Reporting (MDR) system, governed under EU MDR Article 87 and administered through EUDAMED.
What are the reporting timelines for adverse events involving medical devices in the US and EU?
Under FDA 21 CFR Part 803, manufacturers must report deaths or serious injuries within 30 calendar days (or 5 working days for events requiring immediate action). EU MDR timelines are stricter: 2 days for public health threats, 10 days for death or serious deterioration, and 15 days for other serious incidents.
What is the difference between adverse event reporting and post-market surveillance?
Post-market surveillance (PMS) is the broader, ongoing system for collecting and analyzing field data on device performance. Adverse event reporting (AER) is the specific regulatory obligation to notify authorities when a device causes or may cause patient harm. AER is one required output of a functioning PMS system.
What happens if a medical device company fails to report adverse events on time?
Consequences can include FDA warning letters, consent decrees, import alerts, notified body suspension of CE marking, or removal of product from the market. Repeated non-compliance can trigger facility inspections and, in severe cases, criminal liability for company officers.
How do I choose the right adverse event reporting consultant for my medical device company?
Key factors to evaluate:
- Jurisdiction coverage — matches your target markets (FDA, EU MDR, Health Canada, TGA, etc.)
- Device-type experience — relevant background in your specific category (SaMD, IVD, implant, combination device)
- Engagement flexibility — project-based, retainer, or embedded support options
- Demonstrated outcomes — track record in MDR submissions, complaint handling, or audit clearance


