
Compliance missteps carry substantial consequences. TGA cannot refund application fees for applications filed under incorrect device classifications, and incomplete submissions delay market entry by months. US companies accustomed to FDA pathways often underestimate TGA's distinct conformity assessment requirements, which may or may not accept FDA clearance as supporting evidence depending on device class and submission pathway.
This guide profiles the top 10 TGA regulatory consultants who specialize in helping US medical device companies navigate Australian market access in 2026—from pre-submission strategy and device classification to Australian Sponsor coordination and post-market compliance.
TLDR
- TGA requires all medical devices to be listed on the ARTG with an appointed Australian Sponsor before marketing
- Strong TGA consultants understand both FDA and TGA regulatory systems—essential when adapting existing FDA technical documentation for Australian submissions
- Top firms were selected based on verified TGA expertise, US-to-Australia pathway knowledge, and documented client outcomes
- Elexes, Emergo by UL, Freyr Solutions, ProPharma Group, and Arazy Group offer end-to-end TGA support for US companies
Why US Medical Device Companies Need a TGA Consultant for Australian Market Entry
The Australian Sponsor requirement creates an immediate structural hurdle for US manufacturers. Under the Therapeutic Goods Act 1989, overseas manufacturers must appoint an Australian Sponsor — a person or entity with an Australian address — to hold the ARTG entry and assume legal responsibility for the device in Australia.
US companies cannot register directly with TGA. They must either establish an Australian subsidiary, partner with a local entity, or engage a consultant who arranges sponsor services.
Beyond sponsorship, TGA's documentation requirements differ substantially from FDA expectations:
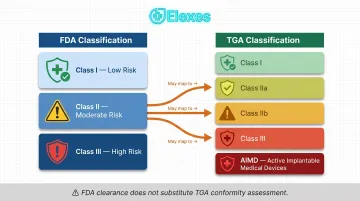
Device classification doesn't map cleanly: TGA uses Classes I, IIa, IIb, III, and AIMD (Active Implantable Medical Devices) — finer risk stratification than FDA's three-class system. An FDA Class II device may land in TGA Class IIa, IIb, or even Class III depending on intended purpose, body contact duration, and active/non-active status.
FDA clearance doesn't substitute for TGA conformity assessment: TGA requires certificates from recognized bodies such as EU Notified Bodies or TGA itself. Per January 2025 guidance, TGA accepts FDA 510(k) Summaries, De Novo Decision Summaries, and PMA decisions as supporting evidence — but these do not bypass TGA's independent assessment. They may reduce the audit scope required, nothing more.
Essential Principles require separate documentation: TGA evaluates devices against Australia-specific Essential Principles that differ from FDA design control requirements. Additional clinical evidence, risk analysis, or technical documentation is often needed to satisfy them.
When evaluating consultants, look for hands-on TGA submission experience, Australian Sponsor network access, and familiarity with how your FDA submission evidence translates — or doesn't — into TGA-acceptable documentation.
Top 10 TGA Regulatory Consultants for US Medical Device Companies (2026)
These firms were selected based on verified TGA submission experience, capacity to serve US-based clients remotely, and documented outcomes in Australian regulatory pathways. Firm details reflect publicly available information verified through official websites and regulatory guidance sources.
1. Elexes
Elexes is a medical device regulatory consulting firm with 50+ years of collective experience, serving 256+ clients across 200+ product types. The firm provides end-to-end TGA regulatory support for US companies — regulatory strategy, technical file preparation, Australian Sponsor coordination, ISO 13485 QMS alignment, and post-market surveillance.
What sets Elexes apart for US companies:
- Cross-regulatory expertise spanning FDA, TGA, EU MDR, and Health Canada — enabling direct repurposing of existing FDA documentation for TGA submissions
- 90% audit clearance rate across global regulatory engagements
- Flexible engagement models (full-time embedded, part-time, or project-based consulting)
- Specialized regulatory due diligence process designed to identify approval "red flags" before submission, reducing rejection risk
Elexes's track record includes successful MDSAP certifications and international registrations across multiple device classes, demonstrating capability with complex, high-risk submissions.
| TGA Services Offered | Device classification, ARTG registration, Australian Sponsor coordination, technical documentation, ISO 13485 QMS support, post-market surveillance |
| Certifications & Credentials | ISO 13485, ISO 9001, ISO 14971, IEC 62304, MDSAP, GCP, GLP |
| Best Suited For | US-based medical device startups, SaMD developers, IVD manufacturers, implant and wearable companies targeting Australian market entry |
2. Emergo by UL
Emergo by UL is a global regulatory consulting firm with a confirmed Australian office at 201 Sussex Street, Sydney NSW 2000. The firm operates 20+ offices across six continents and provides TGA regulatory services through its proprietary RAMS (Regulatory Affairs Management Suite) and OPUS human factors platforms.
Why US companies choose Emergo:
- Physical Australian presence enables direct TGA liaison and local regulatory intelligence
- RAMS platform automates registration management, tracks regulatory changes, and prevents certificate expirations
- ISO-certified quality systems ensure consistent service delivery
- Extensive track record with thousands of devices registered globally across established and emerging markets
Emergo's Sydney office provides in-country expertise that benefits US companies lacking local regulatory infrastructure.
| TGA Services Offered | Market access consulting, ARTG registration, device classification, conformity assessment support, QMS consulting, human factors research |
| Certifications & Credentials | ISO certification (company-wide quality systems), UL certification body affiliation |
| Best Suited For | Mid-to-large US medical device companies seeking established Australian regulatory presence and technology-enabled compliance management |
3. Freyr Solutions
Freyr Solutions maintains a physical Australian office and offers direct TGA sponsor services — a key differentiator for US companies without Australian legal entities. The firm provides end-to-end ARTG registration support, including GMDN code selection, technical file review, post-market surveillance, adverse event reporting, and annual TGA reporting.
Freyr's TGA advantages:
- Direct Australian TGA sponsor service — Freyr can serve as the legal sponsor-of-record
- Freya AI-powered regulatory intelligence chatbot provides instant answers to TGA compliance questions
- 10 Global Delivery Hubs ensure US-Australia collaboration across time zones
- Explicit coverage of all TGA device classes: I (including sterile/measuring), IIa, IIb, III, AIMD, IVDs, and SaMD

Freyr's willingness to act as Australian Sponsor eliminates one of the most significant barriers for US manufacturers entering the Australian market.
| TGA Services Offered | Australian TGA sponsor services, ARTG registration, GMDN code selection, technical file review, post-market surveillance, adverse event reporting, annual TGA reporting |
| Certifications & Credentials | Global regulatory consulting credentials across FDA, EU MDR, TGA, and Health Canada pathways |
| Best Suited For | US medical device startups and mid-sized companies needing direct Australian sponsor services and full ARTG lifecycle support |
4. ProPharma Group
ProPharma Group is a global life sciences regulatory and compliance firm with dedicated Asia-Pacific regional leadership. In August 2023, ProPharma appointed Neama Baho as Associate Vice President, Regulatory Sciences, overseeing regulatory operations across Japan and Asia-Pacific markets. Baho brings 25+ years of experience spanning 60+ markets, including Australia.
ProPharma's TGA differentiators:
- Integrated service model combining regulatory sciences, clinical research, compliance, pharmacovigilance, and medical information
- Asia-Pacific regional team with documented JAPAC market expertise
- Part of The Weinberg Group, providing Washington, DC-based regulatory and compliance capabilities
- Strong cross-functional support for devices requiring parallel regulatory, clinical, and quality workstreams
While ProPharma does not maintain a dedicated Australian office, its Asia-Pacific regional capabilities and integrated service model support complex, multi-jurisdictional submissions including TGA pathways.
| TGA Services Offered | Regulatory sciences, Asia-Pacific market access, regulatory strategy, submission preparation, compliance support (managed through JAPAC regional team) |
| Certifications & Credentials | Global regulatory consulting credentials; Weinberg Group integration provides additional compliance and quality expertise |
| Best Suited For | US pharmaceutical and combination device companies requiring integrated regulatory, clinical, and pharmacovigilance support across Asia-Pacific markets |
5. MCRA (an IQVIA business)
MCRA offers global regulatory consulting across the US, EU, UK, Japan, Canada, and other global markets, with Australia categorized under the broader Canada & Other Global Markets service line. MCRA works with 850+ companies annually on 1,700+ projects, integrating regulatory, clinical, reimbursement, quality assurance, and cybersecurity services.
MCRA's strengths for TGA work:
- Deep therapy-area expertise in neuro-musculoskeletal healthcare, tissue regeneration, and biologics/orthobiologics
- Integration of regulatory and reimbursement consulting — important for understanding Australian market access beyond ARTG registration
- Washington, DC headquarters provides a strong FDA regulatory foundation for US-to-Australia pathway alignment
MCRA does not maintain a TGA-specific service page; Australia capabilities are referenced under broader "global markets" consulting. US companies should confirm TGA-specific experience and resource availability during initial consultations.
| TGA Services Offered | Global regulatory consulting including Australia under "other global markets" designation; likely delivered through partner networks or international regulatory team |
| Certifications & Credentials | IQVIA business unit; global regulatory consulting credentials across FDA, EU MDR, Health Canada, and international pathways |
| Best Suited For | US medical device companies with complex therapy-area requirements (orthopedics, neuromodulation, biologics) seeking integrated regulatory and reimbursement expertise |
6. Arazy Group
Arazy Group delivers technology-first regulatory consulting across 140+ countries using proprietary LICENSALE and REGISLATE platforms. The firm's AI-powered compliance tools generate registration files in under one minute and manage up to 80% of compliance data using previous submission records — reducing labor time and compliance costs by up to 75%.
Arazy's TGA differentiators:
- LICENSALE: Full-service global registration system with expert regulatory guidance for companies needing comprehensive support
- REGISLATE: Self-service registration management software for in-house regulatory teams seeking cost-effective tools
- ISO 9001 certified through October 2026
- 30 years of regulatory experience with 99% market clearance success rate since 2012
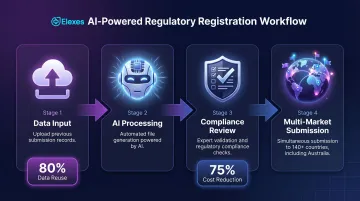
Arazy's platform-based approach suits US companies planning multi-market expansion (including Australia) and seeking scalable, technology-enabled compliance management.
| TGA Services Offered | Device-specific registrations in 140+ countries including Australia; ARTG registration covered within global platform capabilities; AI-powered compliance file generation |
| Certifications & Credentials | ISO 9001:2015 certified (valid through 10/2026); 30 years regulatory expertise across global markets |
| Best Suited For | US medical device companies planning multi-country expansion (including Australia) and seeking technology-enabled, scalable regulatory solutions |
7. Pacific Bridge Medical
Pacific Bridge Medical, founded in 1988 and headquartered in Bethesda, MD, specializes in Asian market regulatory consulting. The firm operates offices in China, Japan, Singapore, Hong Kong, and the United States, with strategic affiliates in India, Indonesia, Korea, Malaysia, Philippines, Taiwan, Thailand, and Vietnam.
Important note: Australia is not listed among Pacific Bridge Medical's offices or strategic affiliate countries on the official website. The firm's documented expertise centers on Asian markets (China, Japan, Korea, Southeast Asia). Official sources don't confirm TGA-specific services.
| TGA Services Offered | Not verified from official website sources; firm focuses on Asian markets (China, Japan, Korea, Southeast Asia) |
| Certifications & Credentials | 37 years of Asia-Pacific regulatory experience; 750+ healthcare companies supported in Asia |
| Best Suited For | US medical device companies prioritizing China, Japan, and Southeast Asian market entry; TGA capabilities should be confirmed directly |
Recommendation: US companies specifically seeking TGA regulatory support should confirm Australian capabilities directly with Pacific Bridge Medical before engagement.
8. MDI Consultants
MDI Consultants is a US-based regulatory consulting firm founded in 1978 and headquartered in Great Neck, New York. The firm specializes in FDA regulatory compliance for medical devices, pharmaceuticals, and food industries, with documented services including FDA 510(k) submissions and Health Canada regulatory submissions.
Important note: No TGA-specific or Australia-specific services were identified on MDI Consultants' official website. The firm's verified expertise centers on FDA and Health Canada submissions. TGA/ARTG registration capabilities could not be confirmed.
| TGA Services Offered | Not verified from official website sources; firm specializes in FDA and Health Canada regulatory pathways |
| Certifications & Credentials | 45+ years FDA regulatory consulting experience |
| Best Suited For | US medical device companies prioritizing FDA and Health Canada submissions; TGA capabilities should be confirmed directly |
Recommendation: US companies specifically seeking TGA regulatory support should confirm Australian capabilities directly with MDI Consultants before engagement.
9. Regulatory Compliance Associates (RCA)
Regulatory Compliance Associates, part of Sotera Health Company, provides regulatory and compliance expert consulting services for life sciences companies. RCA serves medical device, pharmaceutical, and biotech sectors with consulting support through product development and regulatory clearance/approval stages.
Important note: No TGA-specific service page or Australia-specific capabilities were documented on RCA's official website during research. The firm describes itself as a "leading global provider" but does not explicitly detail TGA or Australian medical device registration services.
| TGA Services Offered | Not verified from official website sources; firm offers global regulatory consulting but Australia-specific services not documented |
| Certifications & Credentials | Part of Sotera Health Company; global life sciences consulting credentials |
| Best Suited For | US medical device companies seeking broad regulatory and compliance consulting; TGA capabilities should be confirmed directly |
Recommendation: US companies specifically seeking TGA regulatory support should confirm Australian capabilities and staff resources directly with RCA before engagement.
10. DTS Regulatory Consultants
DTS Regulatory Consultants is an Australian-based TGA regulatory consulting firm located in Brisbane. The firm provides TGA Medical Devices regulatory consulting and ARTG registration support with a team of consultants experienced in TGA Medical Devices and Listed Medicines.
DTS's TGA advantages:
- Brisbane-based firm with direct TGA regulatory authority relationships and in-country presence
- Team includes consultants with hands-on TGA Medical Devices submission experience
- Direct access to Australian regulatory intelligence and TGA liaison capabilities
For US companies, DTS provides the practical advantage of a local Australian partner who knows the TGA's expectations firsthand — without routing through international consulting networks.
| TGA Services Offered | TGA Medical Devices regulatory consulting, ARTG registration support, TGA Listed Medicines consulting |
| Certifications & Credentials | Australian-based regulatory consultants with documented TGA submission experience |
| Best Suited For | US medical device companies seeking a local Australian regulatory partner with direct TGA relationships and in-country expertise |
Contact: Phone: +61 7 3390 8777 | Email: admin@dtsreg.com.au
How We Chose the Best TGA Regulatory Consultants for US Companies
US medical device companies often select consultants based on FDA experience alone — without checking TGA submission track records, Australian Sponsor knowledge, or conformity assessment familiarity. That gap leads to costly delays: misclassified devices, incomplete technical files, and failed sponsor arrangements.
Why TGA-Specific Expertise Matters More Than General International Experience
TGA's classification rules may differ from overseas jurisdictions for identical devices. A device classified as FDA Class II may require reclassification as TGA Class IIa, IIb, or III depending on intended purpose, body contact characteristics, and whether it contains biological materials. TGA explicitly warns that it "cannot refund application fees for applications entered with the wrong class." Verifying classification before submission is not optional — it's the first thing to get right.
Classification risk is only part of the picture. Australian Sponsor responsibilities go well past administrative coordination:
- Ensuring applications are submitted complete with all necessary supporting information
- Responding rapidly to TGA requests for additional information during review
- Maintaining ARTG entries and notifying TGA within 60 days if manufacturer certificates are suspended or revoked
- Reporting adverse events within legislated timeframes
- Maintaining records for up to 10 years (Class IIb implantable and Class III devices)

Consultants without direct TGA experience may underestimate these obligations or fail to establish proper sponsor agreements, jeopardizing post-market compliance.
Key Selection Criteria
We evaluated firms based on the following factors:
TGA-specific service scope:
- ARTG registration support (Class I through Class III and AIMD)
- Conformity assessment guidance and certificate coordination (EU Notified Body recognition, MDSAP acceptance)
- Australian Sponsor coordination or direct sponsor services
- Device classification verification using TGA-specific Classification Rules (Schedule 2, Regulations 3.1-3.3)
US-to-Australia pathway knowledge:
- Understanding of how FDA documentation (510(k), PMA) can be leveraged as supporting evidence under TGA's January 2025 guidance
- Experience aligning FDA technical files with TGA Essential Principles requirements
- Knowledge of when FDA clearance can abridge TGA conformity assessment versus when independent TGA evaluation is required
Certifications and quality systems:
- MDSAP certification (TGA is fully dependent on MDSAP for QMS approval)
- ISO 13485 implementation expertise
- ISO 14971 risk management and IEC 62304 software lifecycle standards
Client track record:
- Verified volume of successful ARTG registrations
- Experience with Class IIb and Class III devices (higher complexity, longer timelines)
- References or case studies demonstrating TGA submission outcomes
Remote service delivery capability:
- Ability to serve US clients without requiring on-site presence
- Flexible engagement models (project-based, part-time, full-time embedded)
- Communication infrastructure for US-Australia time zone coordination
Post-approval compliance support:
- Post-market surveillance (PMS) planning and reporting
- Adverse event reporting and vigilance support
- ARTG entry maintenance and certificate renewal management
Practical Due Diligence Tip
Before signing an engagement, US medical device companies should ask potential TGA consultants:
- How many Class IIb or Class III ARTG registrations have you completed for US-based manufacturers in the past 24 months?
- Can you provide references or case study summaries demonstrating successful TGA submissions?
- Do you provide Australian Sponsor services directly, or do you coordinate with third-party sponsors?
- What is your approach to aligning existing FDA technical documentation with TGA Essential Principles requirements?
- What is your typical timeline and fee structure for ARTG registration support for your specific device class?
Consultants with genuine TGA expertise answer these questions with specific timelines, named submission examples, and clear sponsor arrangements — not vague claims about broad international experience.
Conclusion
Entering the Australian medical device market represents a real strategic opportunity for US companies, but TGA registration demands a different regulatory skill set than FDA compliance. TGA's five-class device system, mandatory Australian Sponsor requirement, and distinct conformity assessment pathways require specialized knowledge that general FDA consulting experience does not provide.
Working with a consultant who understands both FDA and TGA regulatory ecosystems is the most effective way to avoid costly delays, failed submissions, or non-compliant ARTG entries.
The firms profiled above offer varying levels of TGA-specific expertise—from direct Australian sponsor services and in-country regulatory presence to technology-enabled multi-market platforms and integrated regulatory-clinical-reimbursement support.
When evaluating shortlisted consultants, prioritize:
- Current TGA pipeline experience, not just past credentials
- Engagement models (project-based, part-time, embedded) that fit your internal regulatory capacity
- Clear timelines and defined deliverables for each ARTG registration phase
- Verified track record with your specific device class and submission pathway
If your evaluation points toward a firm with cross-regulatory depth and direct TGA experience, Elexes is worth a close look. Elexes supports US medical device companies through every phase of TGA registration—pre-submission strategy, technical file preparation, Australian Sponsor coordination, and post-market surveillance. With 50+ years of collective experience, 90% audit clearance rates, and expertise spanning FDA, TGA, EU MDR, and Health Canada, Elexes helps US manufacturers enter the Australian market efficiently and compliantly.
Ready to explore Australian market entry for your medical device? Contact Elexes at +1 408-475-8091 for a consultation tailored to your device class, regulatory pathway, and market entry timeline.
Frequently Asked Questions
What regulatory agency is TGA?
TGA (Therapeutic Goods Administration) is Australia's federal regulatory body responsible for evaluating and monitoring the safety, quality, and efficacy of therapeutic goods — including medical devices, medicines, and biologicals. It operates under the Therapeutic Goods Act 1989 and is administered by the Department of Health, Disability and Ageing.
What is the difference between MHRA and TGA?
MHRA (Medicines and Healthcare products Regulatory Agency) is the UK's regulatory authority, while TGA is Australia's equivalent. Both regulate medical devices and medicines but operate under separate legal frameworks with different classification systems. A device approved by the MHRA with UKCA marking does not automatically qualify for TGA registration in Australia—independent market authorization is required.
Do US medical device companies need an Australian Sponsor for TGA registration?
Yes. Under TGA rules, US manufacturers cannot hold an ARTG entry directly. They must appoint an Australian Sponsor — a person or company with an Australian address — who becomes legally responsible for the device in Australia. A TGA regulatory consultant can help facilitate or arrange a suitable Sponsor, and some consulting firms are able to serve in that role directly.
How long does TGA registration take for a medical device?
TGA registration timelines vary by device class and pathway. Class I devices can often be listed within 3 working days if documentation is complete. Conformity assessment applications for higher-risk devices average 137 working days, with a statutory maximum of 255 working days. Class III and AIMD devices requiring full TGA conformity assessment typically take 6-12+ months from initial submission to ARTG listing.
Can FDA-cleared devices get expedited TGA approval in Australia?
TGA does not offer an automatic expedited pathway based solely on FDA clearance. FDA-cleared devices can, however, use existing clinical and technical data in TGA submissions. Under TGA's January 2025 guidance, 510(k) Summaries, De Novo Decision Summaries, and PMA decisions are accepted as supporting evidence and may reduce audit scope, but TGA still conducts its own independent evaluation.
What device classification system does TGA use, and how does it differ from FDA classification?
TGA classifies medical devices into Classes I, IIa, IIb, III, and AIMD (Active Implantable Medical Devices) based on risk — a finer stratification than FDA's three-class system. An FDA Class II device may fall into TGA Class IIa, IIb, or even Class III depending on intended purpose, body contact duration, and active/non-active status. US companies should confirm their TGA classification early, as misclassification affects the submission pathway and TGA application fees are non-refundable.


