
Introduction
CAPA management represents the single highest-risk compliance area for medical device manufacturers. According to February 2026 FDA data, CAPA accounts for 12.42% of all FDA 483 observations — holding the #1 position for at least 17 consecutive years. Together with design controls (12.32%) and complaints (10.61%), these three deficiencies account for roughly one-third of all FDA inspection findings.
The stakes are equally high in international markets. Under EU MDR Article 10(9)(h), manufacturers must maintain "corrective and preventive actions and verification of their effectiveness" as an explicit QMS requirement. With the Quality Management System Regulation (QMSR) effective February 2, 2026, every FDA-regulated manufacturer must also align CAPA procedures to ISO 13485:2016 Clauses 8.5.2 and 8.5.3.
Gaps in CAPA compliance don't just trigger Warning Letters. They lead to product recalls, failed certifications, and blocked market access across multiple jurisdictions.
Specialized CAPA management consultants can close those gaps before regulators find them. The right partner brings:
- Device-specific experience across product types and risk classifications
- Regulatory depth spanning FDA, ISO 13485, and EU MDR frameworks
- Demonstrated audit clearance outcomes, not just documentation support
This guide profiles the top 10 consultants to consider in 2026.
TL;DR
- CAPA compliance is mandatory under FDA 21 CFR 820, ISO 13485 Clause 8.5, and EU MDR Article 10 — covering root cause investigation through verified effectiveness
- Weak CAPA systems drive more FDA Warning Letters than any other single deficiency — regulatory exposure is highest here
- Consultants on this list were selected for regulatory expertise, CAPA-specific capabilities, audit clearance rates, and global coverage
- Elexes leads with 50+ years of collective experience, a 90% audit clearance rate, and end-to-end CAPA support across FDA, ISO 13485, and EU MDR
- The right CAPA consultant resolves root causes, clears audits, and strengthens your QMS — without locking you into long-term dependency
What Is CAPA Management and Why Does It Matter for Medical Device Companies?
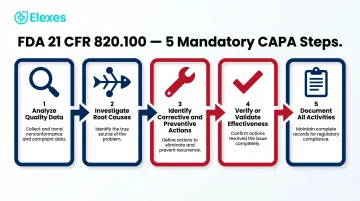
CAPA stands for Corrective and Preventive Action — a formal, documented quality process required under FDA 21 CFR 820.100 and ISO 13485:2016 Clauses 8.5.2 and 8.5.3. The process requires identifying nonconformities, investigating root causes, implementing corrective actions to eliminate problems, applying preventive actions to stop recurrence, and verifying effectiveness before closure.
CAPA is not optional. Under FDA 21 CFR 820.100, manufacturers must establish procedures that:
- Analyze quality data to identify existing and potential causes of nonconforming product
- Investigate the cause of nonconformities related to product, processes, and quality systems
- Identify actions needed to correct and prevent recurrence
- Verify or validate corrective and preventive action effectiveness
- Document all activities
More companies receive FDA Warning Letters for CAPA deficiencies than for any other reason — a pattern that has held for at least 17 years. Failed CAPA systems trigger 483 observations, import alerts, and consent decrees.
CAPA management consulting helps companies address these risks through targeted expertise:
- Design compliant CAPA workflows aligned with FDA and ISO requirements
- Conduct root cause analysis using 5 Whys, fishbone diagrams, and fault tree analysis
- Integrate CAPA with change control and risk management per ISO 14971
- Prepare documentation and processes for FDA inspections and ISO audits
The consultant's role is to build audit-defensible systems that catch nonconformities early and close them effectively.
Top 10 CAPA Management Consultants for Medical Device Companies in 2026
Selection criteria used for this list:
- Regulatory depth in CAPA under FDA 21 CFR 820/QMSR, ISO 13485, and EU MDR
- Documented audit track record and clearance rates
- Service breadth covering root cause analysis, QMS integration, and post-market surveillance
- Measurable, verifiable client outcomes
1. Elexes
Elexes is a medical device regulatory and quality consulting firm with 50+ years of collective experience and a 90% audit clearance rate across 250+ successful projects and 100+ global clients. The firm provides end-to-end CAPA support integrated within broader QMS support across FDA, ISO 13485, EU MDR, and MDSAP-regulated markets.
Differentiators:
- End-to-end CAPA integration within QMS support, ensuring CAPA outputs feed into risk management file updates, clinical evaluations, and design change assessments
- Flexible engagement models: full-time, part-time, or project-based consulting tailored to startup, mid-sized, or established manufacturers
- Global regulatory expertise spanning FDA, Health Canada, EMA, MHRA, and HSA, with local-language auditors for international compliance
- Complete client confidentiality and cross-functional outsourcing combining regulatory, quality, and clinical tasks under one engagement
| Category | Details |
|---|---|
| Key CAPA Services | CAPA workflow design, root cause analysis support, QMS support, ISO 13485 compliance, audit readiness, FDA inspection preparation, post-market surveillance integration |
| Regulatory Coverage | FDA (21 CFR 820/QMSR), ISO 13485, EU MDR, MDSAP, Health Canada, MHRA, EMA |
| Best For | Startups to global medical device companies needing integrated CAPA and QMS support across multiple regulatory markets |

2. Emergo by UL
Emergo by UL is a globally recognized regulatory and quality consulting firm with 400+ professionals across offices in the US, Europe, and Asia, offering comprehensive QMS and CAPA consulting as part of their full lifecycle services. Founded in 1997 and acquired by UL Solutions in 2017, Emergo maintains ISO 13485 certification at the company level and serves clients in more than 20 countries.
Why clients choose them:
- Deep experience across nearly all device categories and regulatory markets, with 25+ global office locations on six continents
- Strong FDA and EU MDR CAPA remediation capabilities backed by UL's testing and certification infrastructure
- Proprietary tools including RAMS (Regulatory Affairs Management Suite) and OPUS (Optimal Product Usability Suite) for streamlined project management
| Category | Details |
|---|---|
| Key CAPA Services | CAPA system implementation, remediation support, ISO 13485 compliance, QMS auditing |
| Regulatory Coverage | FDA, EU MDR/IVDR, global (40+ countries) |
| Best For | Mid-to-large manufacturers needing multi-country CAPA and QMS support |
3. RQM+
RQM+ is a pure-play regulatory and quality consulting firm with a focus on medical devices, diagnostics, and combination products. Headquartered in Cary, NC with a European HQ in Altrincham, UK, RQM+ describes itself as "The MedTech CRO" and maintains ISO 9001 certification.
Differentiators:
- Deep bench of former FDA and Notified Body professionals with insider perspectives on CAPA expectations during inspections
- Particularly strong in CAPA-driven quality system remediation for companies responding to FDA Warning Letters or EU MDR nonconformities
- Emergency support services including CAPA redesign, regulatory narratives, and Notified Body responses
| Category | Details |
|---|---|
| Key CAPA Services | CAPA system remediation, Warning Letter response, MDSAP preparation, ISO 13485 implementation |
| Regulatory Coverage | FDA, EU MDR/IVDR, MDSAP, Japan |
| Best For | Companies with CAPA deficiencies responding to FDA findings or preparing for MDSAP audits |
4. NAMSA
NAMSA is a full-service medical CRO and consulting firm founded in 1967 with 1,400+ scientists, technicians, and consultants across the US, Europe, and Japan. The company serves more than 3,000 medical device and IVD companies annually and has tested nearly 120,000 devices in the past year. NAMSA maintains multiple accreditations including ISO 9001:2015, ISO/IEC 17025:2017, and GLP certification.
Differentiators:
- Unique integrated model combining CAPA consulting with in-house biocompatibility, sterilization validation, and packaging testing
- Handles CAPA investigations that require product testing in-house, removing the delay of coordinating with an external lab
- 50% of all new FDA Premarket Approvals for high-risk medical innovations are entrusted to NAMSA
| Category | Details |
|---|---|
| Key CAPA Services | CAPA investigation support, QMS development, ISO 13485 compliance, regulatory submissions, biological safety consulting |
| Regulatory Coverage | FDA, EU MDR, global |
| Best For | Implantable and high-risk device manufacturers needing CAPA support linked to product testing |
5. Regulatory Compliance Associates (RCA)
RCA is a life sciences consulting firm headquartered in Pleasant Prairie, WI, serving as a Nelson Labs company within the Sotera Health group. The firm serves more than 5,000 customers globally, including 40 of the top 50 medical device companies. RCA maintains ISO 9001 certification and employs former FDA officials.
Standout capabilities:
- Former FDA staff provide insider-level perspective on CAPA expectations during inspections
- Strong track record helping companies respond to 483 observations, Warning Letters, Untitled Letters, and Consent Decrees
- Quality system remediation plans tailored to specific enforcement actions
| Category | Details |
|---|---|
| Key CAPA Services | CAPA remediation, FDA 483 response, mock inspections, quality system development, compliance auditing |
| Regulatory Coverage | FDA (primary), ISO 13485 |
| Best For | US-based manufacturers facing FDA compliance challenges or needing CAPA remediation after inspection findings |
6. Qserve Group
Qserve Group is a specialist medical device and IVD consulting firm headquartered in Arnhem, Netherlands with offices in the US, UK, and China. The firm claims to be "the largest consultant company in the EU that is 100% focused on medical devices and in-vitro diagnostics," with a track record of 750+ successful MDR submissions, 500+ 510(k) approvals, and 100+ PMCF surveys performed.
Differentiators:
- Consultants recruited directly from Notified Body roles bring firsthand knowledge of what assessors expect during EU MDR conformity assessments
- Documented expertise in aligning CAPA systems with Notified Body requirements for EU MDR conformity
- Proprietary tools including Qserve InSight (regulatory compliance) and Qserve Learn (online training)
| Category | Details |
|---|---|
| Key CAPA Services | CAPA system design, EU MDR conformity preparation, ISO 13485 implementation, internal auditing, post-market surveillance |
| Regulatory Coverage | EU MDR/IVDR (primary), FDA, ISO 13485 |
| Best For | EU-focused manufacturers and IVD companies needing CAPA aligned with Notified Body expectations |
7. QES Medical
QES Medical is a Boston-based quality and regulatory consulting firm founded in 2009 by Oscar Garcia, who holds 20 years of medical device industry experience including previous roles at Abbott Laboratories and Covidien (now Medtronic). The firm specializes in hands-on QMS implementation and CAPA support for medical device and pharmaceutical companies.
Notable strengths:
- Practical, build-from-the-ground-up approach to QMS and CAPA systems well-suited for early-stage companies
- Strong in SaMD (Software as Medical Device) and risk management integration with CAPA processes
- Founder-led engagement model ensures consistent quality and senior-level attention
| Category | Details |
|---|---|
| Key CAPA Services | QMS and CAPA system buildout, risk management integration, FDA compliance, ISO 13485, SaMD quality support |
| Regulatory Coverage | FDA, ISO 13485 |
| Best For | Early-stage and startup medical device companies building their first CAPA system |
8. Medpoint
Medpoint is a US-based full-service medical device consultancy with a team of 350 global consultants offering quality assurance, regulatory affairs, clinical support, and technical consulting. The firm maintains ISO 9001:2015 certification and serves medical device, diagnostic, pharmaceutical, and biotech companies.
Differentiators:
- Large team enables rapid scaling across multiple projects with fast resource deployment
- Breadth of services means CAPA management can be coordinated alongside supplier quality audits and clinical support in one engagement
- Global supplier audit program with native language-speaking consultants for international compliance
| Category | Details |
|---|---|
| Key CAPA Services | CAPA support, QMS compliance (21 CFR 820/QMSR), MDSAP readiness, supplier auditing, clinical support |
| Regulatory Coverage | FDA, ISO 13485, MDSAP |
| Best For | Companies needing multi-disciplinary CAPA and QMS support with fast resource scaling |
9. ProPharma Group
ProPharma Group is a large global life sciences consulting firm headquartered in Raleigh, NC with additional offices in the UK and India. The firm serves pharmaceutical, biotech, and medical device companies, with awards including the 2024 CPHI Regulatory and Compliance Award. ProPharma has delivered 20,000+ investigation reports over time.
Differentiators:
- Broad cross-industry expertise particularly valuable for combination product manufacturers where CAPA must align across drug and device quality systems
- Large team allows rapid engagement scaling with project teams up to 45 members
- Tailored CAPA lifecycle management including issue identification, intake documentation, and effectiveness checks
| Category | Details |
|---|---|
| Key CAPA Services | CAPA management, quality investigations, root cause analysis, QMS design, regulatory strategy, combination product quality systems |
| Regulatory Coverage | FDA, EU MDR, global |
| Best For | Combination product manufacturers and large medtech companies with complex CAPA and QMS needs |
10. Rook Quality Systems
Rook Quality Systems is a US-based consulting firm headquartered in Dunwoody, GA, founded in 2012 by Kyle Rose. The firm employs 13 certified quality auditors (CQAs) for FDA 21 CFR 820, ISO 13485:2016, MDSAP, and CE Mark under MDR, and has worked with over 300 MedTech companies.
What sets them apart:
- Team of certified quality auditors with MDSAP, EU MDR CE marking, and ISO 13485 expertise
- Lean, efficient CAPA system designs built specifically for growing companies that need scalable quality infrastructure
- Specializations across Class I–III devices, SaMD, and IVDs
| Category | Details |
|---|---|
| Key CAPA Services | CAPA process development, MDSAP preparation, EU MDR CE marking, ISO 13485 implementation, quality auditing |
| Regulatory Coverage | FDA, ISO 13485, EU MDR, MDSAP |
| Best For | Emerging and growing medical device companies building scalable CAPA and quality systems |
What to Look for in a CAPA Management Consultant
CAPA consulting requires more than general regulatory experience. Look for demonstrated expertise in root cause analysis methodologies — 5 Whys, fishbone/Ishikawa diagrams, and fault tree analysis — alongside CAPA workflow design and effectiveness verification.
The consultant should also understand how CAPA integrates with change control, risk management per ISO 14971, and post-market surveillance. These connections determine whether a CAPA system actually holds up under audit.
Key vetting criteria:
- Ask for audit clearance rates, 483 response outcomes, and anonymized case studies showing measurable CAPA impact
- Verify credentials: RAC-Devices (RAPS), CQE or CQA (ASQ), or ISO 13485 lead auditor certification
- Confirm hands-on experience with your target market — FDA 21 CFR 820/QMSR, ISO 13485, EU MDR, MDSAP, or Health Canada
- Clarify the engagement model (project-based, retainer, or embedded) and match it to your internal team's capacity
Common selection mistakes to avoid:
- Choosing a consultant with no device-specific CAPA experience (pharmaceutical CAPA differs significantly from medical device CAPA)
- Accepting vague scope of work without defined deliverables, timelines, or success criteria
- Selecting based on price alone without assessing depth of regulatory knowledge or audit track record

Beyond written procedures, expect a qualified CAPA consultant to deliver:
- Root cause analysis tools configured for your product type and risk level
- Effectiveness verification protocols with defined acceptance criteria
- CAPA training for your internal quality team
- Integration plans connecting CAPA to your broader QMS
How We Chose the Top 10 CAPA Management Consultants
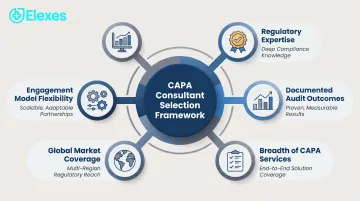
Our evaluation framework prioritized measurable outcomes over brand reputation. Each consultant was assessed on five criteria:
Regulatory Expertise Specific to CAPA
- Direct experience with FDA 21 CFR 820/QMSR, ISO 13485 Clause 8.5, and EU MDR Article 10(9)(h))
- Knowledge of root cause analysis methodologies and effectiveness verification requirements
- Hands-on experience integrating CAPA across change control, risk management, and post-market surveillance
Documented Audit Outcomes
- Audit clearance rates and 483 response success
- Track record helping clients respond to Warning Letters or failed audits
- Testimonials and case studies demonstrating remediation impact
Breadth of CAPA-Related Services
- Root cause analysis facilitation and training
- QMS integration and procedural development
- Post-market surveillance and complaint handling linkage
- Effectiveness verification and closure protocols
Global Market Coverage
- Ability to support FDA, ISO 13485, EU MDR, MDSAP, Health Canada, and other jurisdictions
- Office presence or consultant networks in target regions
- Language capabilities for international audits
Engagement Model Flexibility
- Project-based, retainer, or embedded professional options
- Scalability for startups vs. large manufacturers
- Clear pricing structures and defined deliverables
Firm size and brand recognition were treated as secondary factors throughout. A smaller firm with a 90%+ audit clearance rate and device-specific CAPA depth will consistently outrank a large generalist firm — and the rankings below reflect exactly that logic.
Conclusion
CAPA management is not a compliance checkbox — it's a foundational quality system that regulators scrutinize more closely than any other subsystem. With CAPA accounting for 12.42% of all FDA 483 observations and driving more Warning Letters than any other deficiency, choosing a consultant with proven CAPA depth, device-specific experience, and clear engagement terms is critical for audit readiness and long-term market access.
Before committing to a partner, evaluate potential consultants against your specific CAPA gaps:
- Initial buildout: Do you need to design a CAPA system from scratch for your first FDA or ISO 13485 audit?
- Remediation after findings: Are you responding to 483 observations or Warning Letters requiring immediate corrective action?
- Integration with post-market surveillance: Do you need to link CAPA to complaint handling, vigilance reporting, and PMS data analysis?
- Multi-market alignment: Are you harmonizing CAPA procedures across FDA, ISO 13485, EU MDR, and MDSAP requirements?
The consultant you choose should integrate seamlessly with your existing QMS, provide measurable deliverables, and build internal capability — not create long-term dependency.
Elexes provides CAPA system design, remediation, and ongoing QMS support for medical device companies across global regulatory markets — backed by 50+ years of collective experience, 250+ successful projects, and a 90% audit clearance rate. If you're building a CAPA system from scratch, responding to 483 observations, or aligning procedures across multiple regulatory frameworks, connect with the Elexes team to discuss your specific situation.
Frequently Asked Questions
What is CAPA in service management?
In quality management, CAPA (Corrective and Preventive Action) is a structured process to identify root causes of nonconformities, resolve them through corrective actions, and prevent recurrence through preventive measures. In medical devices, CAPA is mandated under FDA 21 CFR 820 and ISO 13485 Clause 8.5.
What is the best QMS software?
Commonly used eQMS platforms for medical device CAPA management include MasterControl, Greenlight Guru, and Qualio. The best fit depends on your company size, device classification, and target regulatory market. A CAPA consultant can help evaluate options and align the platform to your procedural workflows.
What are the best case management systems?
In medical device quality systems, case management typically covers CAPA and complaint handling workflows. Effective case management requires a compliant eQMS platform paired with procedures aligned to FDA 21 CFR 820 and ISO 13485.
What does CAPA stand for in medical devices?
CAPA stands for Corrective and Preventive Action. Corrective Action eliminates the root cause of an existing nonconformity, while Preventive Action addresses potential issues before they occur. Regulators expect your QMS to document both, verify their effectiveness, and integrate them into ongoing quality oversight.
When should a CAPA be initiated in a medical device company?
Initiate a CAPA when quality data — complaints, audit findings, post-market surveillance signals, or nonconformance reports — points to a systemic issue or patient safety risk. Not opening a CAPA when the evidence warrants one is itself a frequent FDA 483 observation.
How do I choose a CAPA management consultant for my medical device company?
Prioritize consultants with device-specific CAPA experience, demonstrated audit clearance outcomes, familiarity with your target regulatory market (FDA, ISO 13485, EU MDR), and a clear scope of work that includes root cause analysis support, effectiveness verification, and QMS integration — not just document-writing services.


