
Introduction
MDSAP compliance isn't optional for medical device companies targeting North American and global markets—it's a business imperative. A single MDSAP audit now covers five regulatory bodies (FDA, Health Canada, ANVISA, TGA, PMDA), eliminating redundant inspections while opening doors to the world's most important med-tech markets. But choosing the wrong consultant can derail the entire process—triggering high-grade non-conformities, regulatory notifications, and months of expensive rework.
Not all MDSAP consultants deliver equal results. Companies frequently struggle with non-conformity grading, documentation gaps, and audit readiness—particularly in purchasing controls and CAPA chapters, which account for the majority of major findings.
Each consultant on this list was evaluated on audit clearance track records, multi-jurisdiction regulatory depth, and demonstrated QMS alignment across FDA, Health Canada, and ANVISA requirements. Here are the ten most reliable MDSAP audit consulting partners in North America for 2026.
TL;DR
- MDSAP is a single-audit program accepted by the US, Canada, Brazil, Australia, and Japan, covering all five markets in one audit cycle
- Evaluate consultants on audit clearance rates, multi-jurisdiction expertise, QMS alignment depth, and MDSAP lead auditor credentials
- The 10 firms listed were selected based on MDSAP-specific experience, North American market presence, client outcomes, and QMS depth
- Elexes leads with a 90% audit clearance rate and 50+ years of collective regulatory experience across MDSAP, ISO 13485, and global QMS compliance
- Match your shortlist to your device type, company size, and target market using the selection criteria section below
What Is MDSAP and Why Does It Matter for North American Manufacturers
MDSAP is a program developed by the International Medical Device Regulators Forum (IMDRF) that allows a single audit of a medical device manufacturer's QMS to satisfy the regulatory requirements of all five participating countries: the US FDA, Health Canada, Brazil's ANVISA, Australia's TGA, and Japan's MHLW/PMDA. Since January 2019, Health Canada has mandated MDSAP as the only recognized QMS certification program for medical device license holders.
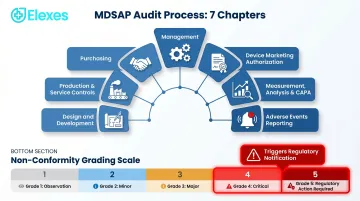
MDSAP audits evaluate seven core process chapters defined in MDSAP AU P0002.009:
- Management
- Device Marketing Authorization
- Measurement, Analysis & Improvement (including CAPA)
- Medical Device Adverse Events Reporting
- Design and Development
- Production and Service Controls
- Purchasing
Non-conformities are graded 1–5 per the GHTF/SG3/N19:2012 grading system. Grade 4 or 5 findings trigger immediate regulatory notification across all five jurisdictions.
For manufacturers targeting Canada, the US, or any of the other three MDSAP jurisdictions, working with an experienced consultant—covering QMS gap assessment, mock audits, and post-audit CAPA—can be the difference between a clean audit and a Grade 4 finding that reaches every regulator at once. The firms below represent the top 10 MDSAP consulting practices serving North American manufacturers.
Top 10 MDSAP Audit Consultants in North America 2026
This list is compiled using publicly available information, evaluated across five criteria:
- MDSAP audit success rates and documented outcomes
- QMS implementation depth and remediation capability
- Multi-jurisdiction regulatory expertise (US, Canada, Brazil, Australia, Japan)
- North American client base and industry reputation
- Flexibility and fit across company sizes and device categories
Elexes
Elexes is a medical device regulatory and quality consulting firm with 50+ years of collective experience and clients across 25+ countries. With a 90% audit clearance rate across 250+ completed projects, the firm offers end-to-end MDSAP readiness — from initial gap assessment through post-audit remediation — with flexible engagement models suited to both startups and mid-size manufacturers.
What sets Elexes apart:
- 90% audit clearance rate across 250+ completed projects
- Multi-standard expertise (ISO 13485, ISO 14971, IEC 62304, MDSAP)
- Flexible engagement models (project-based, part-time, or full outsourcing)
- Complete data confidentiality and transparent deliverables
- Strong fit for startups and mid-size device companies needing end-to-end MDSAP readiness support
| Core MDSAP Services | Key Certifications / Expertise | Best For |
|---|---|---|
| QMS gap analysis, mock MDSAP audits, ISO 13485 implementation, CAPA management, post-audit support | MDSAP, ISO 13485, ISO 14971, IEC 62304, FDA QSR/QMSR, EU MDR | Startups to mid-size device companies seeking full MDSAP readiness with flexible engagement and global regulatory coverage |

Emergo by UL
Emergo by UL (headquartered in Austin, TX, part of UL since 2019) brings 400+ professionals and offices across North America, Europe, and Asia-Pacific. MDSAP preparation sits within a full-lifecycle quality and regulatory portfolio covering 40+ markets — making it a strong option for manufacturers managing MDSAP alongside global market access.
Key differentiators:
- Deep bench across 40+ regulatory markets
- Proprietary RAMS® platform for regulatory affairs management
- Strong FDA and ISO 13485 implementation track record
- Capability to bundle MDSAP prep with global market access strategy
| Core MDSAP Services | Key Certifications / Expertise | Best For |
|---|---|---|
| MDSAP audit prep, ISO 13485 implementation, QMS consulting, internal audits, global registration support | ISO 13485, MDSAP, FDA QMSR, EU MDR/IVDR, global regulatory representation | Mid-size to large manufacturers seeking integrated MDSAP + global market access under one firm |
RQM+
RQM+ (formed in 2020 from a merger of regulatory consulting firms, headquartered in Cary, NC) is a pure-play life sciences regulatory and quality firm with 400+ consultants and offices in the US, UK, Germany, Netherlands, and Japan. MDSAP readiness is a core service alongside FDA QMSR transition and EU MDR compliance.
Key differentiators:
- Large team of former FDA and Notified Body professionals
- Strong quality system remediation capability ideal for companies with prior audit findings
- MDSAP-specific audit preparation aligned with current grading criteria
| Core MDSAP Services | Key Certifications / Expertise | Best For |
|---|---|---|
| MDSAP prep, QMS remediation, mock inspections, ISO 13485 support, FDA QMSR transition | ISO 13485, MDSAP, FDA QMSR, EU MDR/IVDR, internal auditing | Companies with prior audit findings or quality system gaps preparing for their first or next MDSAP audit |
Medpoint
Medpoint (US-based, 350 global consultants) is a full-service consultancy with MDSAP compliance support as a core service. The firm covers QA, regulatory, clinical, and global supplier auditing — with multilingual auditors and international reach supporting North American manufacturers managing complex supply chains.
Key differentiators:
- Large consultant pool enables rapid resourcing for MDSAP readiness projects
- Strong global supplier audit capabilities relevant to MDSAP's purchasing controls chapter
- Broad device category experience
| Core MDSAP Services | Key Certifications / Expertise | Best For |
|---|---|---|
| MDSAP compliance support, QA consulting, supplier auditing, FDA 21 CFR 820/QMSR compliance, clinical support | ISO 13485, MDSAP, FDA QSR/QMSR, global supplier auditing | Manufacturers needing MDSAP prep plus supplier audit support, especially multi-site or global supply chain companies |

mdiConsultants
mdiConsultants (headquartered in New York, founded by a former FDA investigator) is a firm with direct hands-on MDSAP audit experience. The company has published detailed case studies on MDSAP certification audits and has helped clients navigate the full multi-country audit process including document preparation, audit schedule logistics, and close-out CAPA.
Key differentiators:
- First-hand MDSAP audit experience from the ground up (including pilot-stage)
- FDA investigator background providing a unique lens on audit intensity differences
- Expertise across US, Canada, Brazil, and Australia regulatory requirements covered under MDSAP
| Core MDSAP Services | Key Certifications / Expertise | Best For |
|---|---|---|
| MDSAP certification prep, Health Canada CMDCAS compliance, FDA compliance audits, ISO compliance, CAPA support | MDSAP, ISO 13485, FDA QSR, Health Canada CMDCAS, CE Mark support | North American manufacturers seeking MDSAP prep from consultants with direct first-hand MDSAP audit participation experience |
Rook Quality Systems
Rook Quality Systems (US-based, 12+ years in operation) is a quality and regulatory consulting firm with MDSAP competency alongside ISO 13485 and EU MDR. The team of 36+ includes 13 certified quality auditors serving hardware, SaMD, and IVD companies from Class I through Class III.
Key differentiators:
- Lean QMS design philosophy suited for smaller device companies and startups


