
Introduction
Medical device classification under EU MDR is the foundational step to accessing the European market. Get it wrong, and every regulatory requirement that follows is built on a cracked foundation.
Manufacturers who misclassify their devices face Notified Body disputes, full rework of technical documentation, and CE marking delays of 6–12 months or more. The consequences reach further than most teams anticipate until they're already in the middle of them.
This article is a practical guide to the EU MDR classification system — covering the 22-rule framework, how to apply classification criteria to your specific device, and the common pitfalls that lead to costly reclassification disputes.
TL;DR
- EU MDR classifies medical devices into four risk-based classes (I, IIa, IIb, III) using 22 rules in Annex VIII
- Your device's class determines the conformity assessment route, whether a Notified Body is required, and the level of clinical evidence needed
- Software (SaMD/MDSW) follows Rule 11 with classifications from Class I to III based on clinical risk severity
- Accessories, nanomaterials, and Annex XVI products follow special classification rules — and often land in a higher class than manufacturers expect
- Prior MDD classification does not carry over — manufacturers must reclassify from scratch under EU MDR
What Is EU MDR Medical Device Classification?
Medical device classification is the process by which a manufacturer assigns a risk class to their device using the 22 classification rules in Annex VIII of Regulation (EU) 2017/745, based on the device's specific intended purpose. That assigned class directly determines whether a Notified Body must be involved, which conformity assessment annexes apply, what clinical evidence is required, and how stringent post-market surveillance obligations will be.
EU MDR replaced the Medical Device Directive (MDD/93/42/EEC) on 26 May 2021, introducing revised classification rules for software, nanomaterials, and combination devices. Regulation (EU) 2017/745 also repealed the Active Implantable Medical Devices Directive, creating a unified regulatory framework across the EU.
Manufacturers must never assume their previous MDD class remains valid — reclassification from scratch under Annex VIII is required.
Understanding what triggers classification starts with the legal definition. Article 2(1) covers any instrument, apparatus, software, implant, or material intended for diagnosis, prevention, monitoring, treatment, or alleviation of disease or disability. Classification applies to any product meeting this definition, regardless of technological complexity or commercial maturity.
The Four EU MDR Device Classes Explained
EU MDR divides all medical devices into Class I, IIa, IIb, and III, where each step up represents greater potential harm and more rigorous regulatory oversight. Your device's class determines Notified Body involvement, clinical evidence requirements, and post-market surveillance obligations — making correct classification the foundation of your EU market strategy.
Class I
Class I represents the lowest-risk category, covering devices with minimal or no direct patient contact risk — such as stethoscopes, walking aids, bandages, and non-sterile examination gloves. In most cases, the manufacturer can self-certify without Notified Body involvement.
Three Class I sub-classifications require Notified Body involvement:
- Class Is: Devices placed on the market in sterile condition
- Class Im: Devices with a measuring function where inaccuracy could affect patient safety
- Class Ir: Reusable surgical instruments requiring reprocessing validation
Class IIa
Class IIa covers medium-risk devices, generally those with short-term body contact (60 minutes to 30 days) or moderate diagnostic and therapeutic impact. Examples include catheters, hearing aids, short-term corrective contact lenses, and dental fillings.
Manufacturers must meet the following requirements:
- Notified Body conformity assessment under Annex IX or XI
- Formal EU Declaration of Conformity
- Post-market surveillance report (PSUR) updated at least every two years
Class IIb
Class IIb covers medium-to-high-risk devices where the potential for serious harm is elevated. Examples include ventilators, insulin pens, long-term corrective contact lenses, incubators, and CT imaging devices.
Class IIb carries stricter obligations than Class IIa:
- Notified Body assessment under Annex IX or XI is mandatory
- Annual Periodic Safety Update Report (PSUR) required
- For implantable Class IIb devices (with certain exceptions), the Notified Body must assess technical documentation per individual device
- Manufacturers of implantable devices must publish a Summary of Safety and Clinical Performance (SSCP) in EUDAMED
Class III
Class III represents the highest-risk class, covering devices that have direct, long-term, or irreversible contact with critical anatomical systems — including pacemakers, prosthetic heart valves, coronary stents, breast implants, surgical meshes, and spinal disc replacements.
Class III carries the most demanding regulatory obligations under EU MDR:
- Conformity assessment via Annex IX or Annex X coupled with Annex XI
- Pre-market clinical investigation is generally compulsory
- Manufacturers must publish a Summary of Safety and Clinical Performance (SSCP) in EUDAMED
- Expert panel consultation applies for Class III implantable devices per Article 54

How the 22-Rule Classification System Works
Annex VIII of EU MDR contains 22 classification rules organized into four categories:
- Rules 1–4: Non-invasive devices
- Rules 5–8: Invasive devices
- Rules 9–13: Active devices
- Rules 14–22: Special rules covering devices such as those incorporating medicinal substances, nanomaterials, contraceptive devices, and software
Duration-of-Use Definitions
Three duration-of-use definitions determine which sub-rules apply:
- Transient: Continuous use less than 60 minutes
- Short Term: 60 minutes to 30 days
- Long Term: More than 30 days
"Continuous use" includes immediate replacement by the same type of device — not just uninterrupted single-session use. This distinction directly affects which duration tier, and therefore which sub-rules, apply to your device.
The Highest Classification Wins
Annex VIII Implementing Rule 3.5 mandates that when multiple rules apply to a single device, the strictest rule resulting in the highest classification must be used.
Take a wound drainage system as an example: the cannula qualifies as Class IIa under Rule 7, while both the tubing and collector qualify as Class I under Rule 1. Assessed as a complete system, the entire device is classified at Class IIa.
Rule 11: Software Classification
Medical device software (SaMD/MDSW) is classified based on the severity of harm its diagnostic or therapeutic output could cause:
- Class I: General-purpose software where decisions carry minimal clinical impact
- Class IIa: Diagnostic decision-support tools where incorrect output could influence treatment decisions
- Class IIb: Software where incorrect output could cause serious deterioration or require surgical intervention
- Class III: Software where failure could result in death or irreversible harm (such as AI stroke diagnosis tools)

MDCG 2019-11 (revised June 2025) provides detailed criteria for MDSW qualification and classification, including clarifications on modules and Rule 11 interpretation.
Step-by-Step Classification Process
Follow this systematic approach:
- Confirm the product qualifies as a medical device under Article 2(1)
- Define the intended purpose with precision, including limitations
- Evaluate all 22 rules systematically — don't stop at the first match
- Apply the strictest applicable rule if multiple rules apply
- Document the full classification rationale in technical documentation as required by Annex II, Section 1.1(f)
Why Getting Classification Right Matters
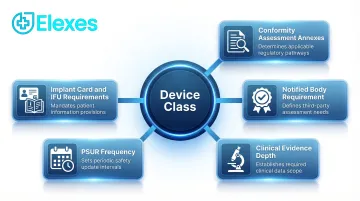
The device class drives the entire regulatory pathway. It determines:
- Which conformity assessment annexes apply
- Whether a Notified Body is required
- The depth of clinical evidence needed
- PSUR frequency and UDI traceability obligations
- Implant card requirements and whether instructions for use are mandatory
The cost and timeline risk of misclassification is substantial. If a manufacturer underclassifies their device and a Notified Body or competent authority challenges it, the dispute must be referred to the national competent authority under Article 51(2). This process can add months to your timeline and require complete rework of technical documentation. The European Commission acknowledged Notified Body capacity constraints during the MDD-to-MDR transition, leading to Regulation (EU) 2023/607 extending transition deadlines — a sign that classification and conformity assessment timelines remain unpredictable.
Given these stakes, determining the correct classification before committing to a conformity assessment route is essential. Elexes has supported 250+ medical device projects across 200+ product types, helping manufacturers resolve classification questions early and build technical documentation that holds up under Notified Body scrutiny.
Special Cases and Common Classification Pitfalls
Special Classification Cases
Medical Device Software (MDSW/SaMD): Software is assessed under Rule 11 using MDCG 2019-11 guidance. Image management systems used only for viewing, archiving, and transmitting images are not medical devices, but systems with post-processing functions for diagnostic purposes qualify as MDSW.
Accessories: Accessories must be classified "in their own right" per Implementing Rule 3.2, separately from the device with which they are used. Do not assume an accessory shares its parent device's class.
Annex XVI Products: Six product categories without an intended medical purpose fall under EU MDR via Annex XVI:
- Contact lenses or items for the eye
- Products for surgically invasive anatomy modification
- Dermal or mucous membrane fillers
- Adipose tissue reduction equipment (liposuction)
- High-intensity electromagnetic radiation equipment (lasers, IPL)
- Brain stimulation equipment
Custom-Made Devices (CMDs): Classification is still required even though CE marking requirements differ from standard devices. Class III custom-made implantable devices require Notified Body involvement under Annex IX or XI.
Common Classification Mistakes
These special cases are also a frequent source of classification errors. Watch for three mistakes in particular:
1. Assuming prior MDD classification still applies: The MDD used 18 rules; EU MDR uses 22. New rules for software (Rule 11), nanomaterials (Rule 19), and closed-loop systems (Rule 22) were added. Always reclassify from scratch under EU MDR.
2. Applying rules based on typical use rather than intended purpose: Classification depends on the manufacturer's stated intended purpose, not how clinicians commonly use the device in practice. Your technical documentation must reflect your actual intended purpose.
3. Failing to evaluate all 22 rules: Missing a special rule — such as Rule 14 for devices incorporating medicinal substances, Rule 18 for nanomaterials, or Rule 22 for active therapeutic devices — can result in an incorrect class. Evaluate every rule before finalizing your classification.

Borderline and Ambiguous Cases
When it's unclear whether a product qualifies as a medical device, IVD, or medicinal product, two options are available. Manufacturers can consult the Manual on Borderline and Classification (4th edition, September 2025) for published guidance, or submit a formal classification inquiry to the national competent authority of their registered place of business.
Conclusion
EU MDR medical device classification is a structured, rules-based process with four risk classes that each unlock a distinct regulatory pathway. Accuracy at this step shapes everything from Notified Body involvement and clinical evidence requirements to post-market surveillance obligations and CE marking timelines.
Annex VIII is complex — and the new EU MDR rules for software, nanomaterials, and combination devices add layers that can push a device into a higher class than expected. Misclassification at this stage means the wrong conformity route, delayed CE marking, and potential market access gaps.
Elexes supports manufacturers through the full classification process, from Annex VIII rule analysis and Notified Body selection through clinical evaluation, post-market surveillance, and CE marking. With 250+ completed projects across 200+ product types, the team brings direct experience with the classification decisions that regulators scrutinize most.
Frequently Asked Questions
How many medical device classifications are in the EU?
There are four main risk classes — Class I, IIa, IIb, and III — defined in Annex VIII of EU MDR 2017/745. Class I is further divided into three sub-classes (Is, Im, Ir) based on sterility, measuring function, or reusability.
What is the difference between Class I, II, and III medical devices?
Class I is low risk with possible self-certification. Class IIa and IIb are medium and medium-to-high risk requiring Notified Body involvement. Class III is the highest risk class, requiring full Notified Body assessment, pre-market clinical investigation, and rigorous post-market surveillance.
What types of devices fall under Class IIa and IIb?
Class IIa covers moderate-risk devices like catheters, hearing aids, and short-term contact lenses. Class IIb includes higher-risk devices such as ventilators, CT scanners, and long-term contact lenses. Both classes require Notified Body involvement, but Class IIb typically demands a more comprehensive technical file and clinical evidence.
How are medical devices approved in the EU?
Medical devices must obtain CE marking by completing a conformity assessment under EU MDR. The required pathway depends on device class, ranging from self-certification for standard Class I devices to mandatory Notified Body assessment and clinical investigation for Class IIa, IIb, and III devices.
What is a Class III CE marked medical device?
Class III devices are the highest-risk category under EU MDR — implantable and life-sustaining devices with the greatest potential patient risk. CE marking requires a Notified Body conformity assessment under Annex IX or X (coupled with Annex XI), and pre-market clinical investigation is required in most cases unless a valid exemption applies.
What is the difference between EU MDR and IVDR?
EU MDR (2017/745) covers medical devices intended for diagnosis, treatment, or monitoring. IVDR (2017/746) covers in vitro diagnostic devices. Both use risk-based classification, but IVDR uses its own four-class system (Class A through D) with different rules that do not map directly to EU MDR's Class I–III structure.


