
Introduction
A failed notified body surveillance audit. A lapsed CE certificate. A Clinical Evaluation Report that no longer reflects real-world device performance. These are the consequences manufacturers face when Post-Market Clinical Follow-Up (PMCF) is treated as an afterthought. Despite being a mandatory requirement under EU MDR, PMCF is consistently misunderstood and poorly executed across the industry. Manufacturers often confuse it with routine complaint handling, treat it as a one-time checkbox at CE marking, or submit generic plans that fail to address device-specific clinical questions.
PMCF is a continuous, proactive process under EU MDR in which manufacturers systematically collect and evaluate clinical data from CE-marked devices already on the market. Its purpose is to confirm ongoing safety and performance, ensure the benefit-risk ratio remains acceptable, and detect emerging risks throughout the device's lifecycle.
Without a functioning PMCF process, your Clinical Evaluation Report becomes outdated — and your certificate faces real suspension risk during notified body surveillance.
That's exactly what this guide addresses. It's written for medical device manufacturers, regulatory teams, quality professionals, and startups navigating EU MDR compliance. We cover what PMCF involves, how the process works from plan to report, which data collection methods are acceptable, and — critically — where manufacturers most commonly go wrong.
TL;DR
- PMCF continuously updates the clinical evaluation of CE-marked devices throughout their lifecycle—not a one-time audit
- Requires a formal PMCF Plan, execution of data collection, and a PMCF Evaluation Report feeding back into the CER
- Data collection methods range from literature reviews and user feedback to device registries and dedicated clinical studies
- Applies to all CE-marked devices, with more rigorous requirements for Class IIb and III devices
- Claiming "not applicable" requires documented justification — and notified bodies scrutinize these claims closely
What Is Post-Market Clinical Follow-Up (PMCF)?
EU MDR Annex XIV, Part B, Section 5 defines PMCF as "a continuous process that updates the clinical evaluation" requiring manufacturers to "proactively collect and evaluate clinical data from the use in or on humans of a device which bears the CE marking." (EU MDR Annex XIV)
In practice, PMCF ensures that the clinical evidence behind your CE marking stays valid as the device is used in real-world conditions across its full lifecycle. The goal is systematic confirmation that your device continues to perform safely as intended — not a one-time data collection exercise.
PMCF vs. Post-Market Surveillance (PMS)
These two obligations overlap but serve distinct purposes:
- PMS covers all post-market activities — vigilance, complaint handling, trend reporting, and field safety corrective actions
- PMCF is the clinical data-focused subset specifically designed to update your Clinical Evaluation Report (CER)
MDR 2025-10 clarifies this relationship: "The PMCF/PMPF plan is an integral part of the PMS plan." (MDCG 2025-10) While PMS data informs corrective actions, PMCF findings specifically feed back into the clinical evaluation.
For IVD manufacturers: The IVDR equivalent is Post-Market Performance Follow-Up (PMPF) under Regulation (EU) 2017/746 Annex XIII, Part B, which feeds into the Performance Evaluation Report (PER) rather than a CER.
Why PMCF Is a Mandatory Regulatory Requirement
Under EU MDR, every manufacturer with a CER must have a PMCF plan. This marks a sharp departure from the predecessor Medical Device Directive (MDD), where PMCF could be easily waived. Justification for not conducting PMCF is now held to a much higher standard.
What EU MDR Demands
Article 61(11) states: "The clinical evaluation and its documentation shall be updated throughout the life cycle of the device concerned with clinical data obtained from the implementation of the manufacturer's PMCF plan... For class III devices and implantable devices, the PMCF evaluation report shall be updated at least annually." (EU MDR Article 61(11))
Key requirements:
- All devices: Lifecycle updating of clinical evaluation using PMCF data
- Class III and implantables: Minimum annual PMCF Evaluation Report updates
- Lower-risk devices: Update frequency must be justified and risk-proportionate
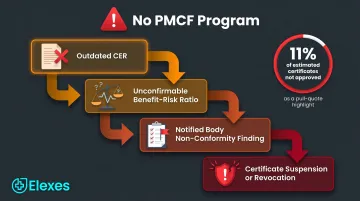
Consequences of Absent PMCF
Without proper PMCF:
- Your CER becomes outdated and no longer reflects real-world device performance
- The benefit-risk ratio can no longer be confirmed with current evidence
- Non-conformity findings arise during notified body surveillance audits
- Certificate suspension or revocation becomes a real risk
A peer-reviewed survey of 13 European Notified Bodies published in February 2025 found that 327 applications (11% of estimated certificates) were not approved, with PMCF studies required as certificate conditions for novel technologies. One case led to certificate cancellation. (Dobrzynska et al., Frontiers in Medical Technology, 2025)
Regulatory Guidance Framework
These consequences are avoidable — but only if your PMCF program aligns with what notified bodies actually expect. MDCG guidance documents provide the templates they use as benchmarks:
- MDCG 2020-7: PMCF Plan Template
- MDCG 2020-8: PMCF Evaluation Report Template
- MDCG 2025-10: Post-market surveillance guidance defining "proactive" data collection obligations
These documents clarify that "proactive" means actively seeking information, not merely waiting for complaints to arrive. (MDCG 2025-10) In practice, that means structured literature reviews, registry participation, and targeted patient/clinician feedback — not passive complaint monitoring.
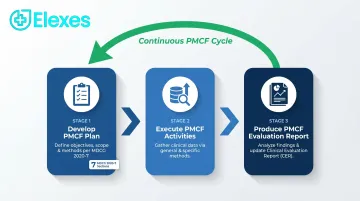
How the PMCF Process Works
The PMCF process flows from identifying clinical data gaps through executing activities and feeding conclusions back into your clinical evaluation and risk management files.
Key inputs include:
- Current CER and identified knowledge gaps
- Risk management file with residual risks
- Existing clinical evidence base
- Post-market complaint data
- Identified performance uncertainties
Key principle: PMCF activities should be targeted at answering specific clinical questions, not gathering data without a clear purpose. Every planned activity should address a defined evidence gap.
Step 1: Develop the PMCF Plan
MDCG 2020-7 outlines seven sections for the PMCF Plan:
| Section | Content |
|---|---|
| A | Manufacturer contact details |
| B | Device description, intended purpose, classification |
| C | Planned general and specific activities (substantive core) |
| D | References to relevant technical documentation |
| E | Evaluation of data from equivalent/similar devices |
| F | Applicable standards and guidance references |
| G | Estimated date of PMCF Evaluation Report |
Section C is the substantive core and must be device-specific and justified. Generic statements like "we will monitor literature" without methodology fail notified body scrutiny.
This requirement is especially strict for higher-risk devices. MDCG 2020-7 states: "For implantable devices and class III devices where clinical investigations have not been performed... the PMCF plan shall include post market studies to confirm the safety and performance of the device." (MDCG 2020-7)
Step 2: Execute PMCF Activities
Each activity must be executed exactly as documented in the plan — deviations require documented justification. Outcomes are recorded against the defined evidence gap each activity was designed to address.
For general activities:
- Literature searches must meet systematic review standards
- Search terms, databases, date ranges, and inclusion/exclusion criteria must be documented
- Results must be analyzed for clinical significance
For specific activities:
- Studies require appropriate design, ethics approval, and data integrity standards
- Registry evaluations need defined endpoints and data collection protocols
- Patient surveys require statistically valid sample sizes and clinical endpoints
Step 3: Produce the PMCF Evaluation Report
The PMCF Evaluation Report follows MDCG 2020-8 structure and records:
- Results of all planned activities
- Clinical significance of findings
- Impact on device safety and performance conclusions
- How findings affect the CER
- Whether corrective actions or plan revisions are needed
The CER feedback loop is mandatory. MDCG 2020-8 requires that "the conclusions of the PMCF evaluation report shall be taken into account to update eventually the clinical evaluation." Section E goes further, specifically mandating that "analysis of the outcome is to be reported in the updated clinical evaluation report." (MDCG 2020-8)
In practice, this means your updated CER must reference PMCF findings directly — notified bodies will look for this traceability.
PMCF Data Collection Methods: General vs. Specific
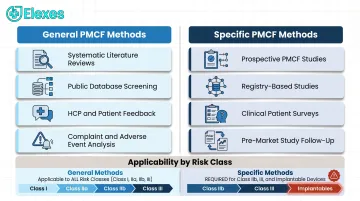
EU MDR Annex XIV, Part B formally separates general and specific methods and requires rationale for your chosen approach.
The Regulatory Distinction
Annex XIV Part B Section 6.2 specifies the PMCF plan shall include:
- (a) General methods: Gathering clinical experience, user feedback, literature screening, and other clinical data sources
- (b) Specific methods: Registry evaluations and PMCF studies
- (c) Rationale: Justification for why chosen methods are appropriate
General PMCF Methods
General methods gather broadly available clinical experience:
- Systematic literature reviews on your device or equivalent devices
- Public database screening (EUDAMED, MAUDE, national registries)
- Structured feedback from healthcare professionals and patients
- Complaint and adverse event analysis with clinical evaluation
For higher-risk devices, general methods alone rarely satisfy notified bodies. As MDCG 2025-10 states, methods "should be appropriate and based on the device's risk profile" — which typically means pairing general methods with targeted specific activities.
Specific PMCF Methods
Specific methods involve more controlled or targeted data collection:
- Prospective PMCF studies (observational or interventional)
- Registry-based studies using real-world data
- Device-specific patient surveys with quantifiable clinical endpoints and statistically valid sample sizes
- Controlled follow-up of pre-market study participants
Survey Quality Threshold
Not all surveys are equal. High-quality surveys with statistically calculated sample sizes and clinical endpoints count as specific methods. Retrospective, open-ended usability questionnaires fall under general methods.
BSI's Head of Clinical Compliance stated in March 2024 that "selected methods should be justified, surveys for example may only be appropriate for lower risk devices, or established technologies." (BSI CORE-MD Presentation) This confirms that notified bodies actively scrutinize method selection.
When Specific Activities Are Required vs. Waivable
Specific activities can be reasonably waived for:
- Low-risk devices with well-established technologies
- No new risks identified
- Sufficient pre-existing clinical data with no long-term performance uncertainties
When neither condition clearly applies, the burden of justification falls on the manufacturer — and that justification must be specific.
Specific activities are expected for:
- Novel technologies
- Implantables and Class III devices
- Devices with residual risks
- Insufficient pre-market clinical data
Critical guidance from MDCG 2020-7: For implantable and Class III devices where clinical investigations were not performed, post-market studies are mandatory. Justifications citing "sufficient pre-market data" without specifying data volume, recency, or gap analysis will be challenged during technical file review.
Elexes works with manufacturers to build PMCF plans that match method selection to device risk class — including written rationale that holds up under notified body scrutiny.
Common Misconceptions and When PMCF May Not Apply
Most Damaging Misconception
"PMCF only applies when a clinical study is needed."
Many manufacturers justify non-applicability by arguing they don't need a specific study. This ignores the mandatory nature of general PMCF activities—literature review and user feedback are expected regardless of risk class whenever a CER exists.
How Teams Misuse the Process
Common mistakes include:
- Treating PMCF as a checkbox completed once at CE marking rather than an ongoing obligation
- Submitting generic plans that aren't device-specific or don't address clinical data gaps
- Using PMCF interchangeably with PMS without explaining how findings update the CER
- Substituting complaint logs for PMCF Evaluation Reports: complaint management is part of PMS, not a replacement for clinical evaluation of post-market data
Limited Circumstances Where PMCF May Not Apply
PMCF is not required when:
- No clinical evaluation report exists (extremely rare under MDR)
- All clinical data gaps have been fully resolved with rigorous documentation
- Residual risks are negligible with no long-term performance uncertainties
Critical caveat: The justification must be specific and technical, not a blanket statement like "sufficient pre-market evidence exists."
Annex II Section 6.1(d) and Annex III Section 1(b) both require either a PMCF plan or "a justification why a PMCF is not applicable." (EU MDR Annex II & III)
MDCG 2020-7 confirms that "the notified body's assessment of the clinical evaluation shall also cover... the justification in relation to non-performance of PMCF."
Regulators and notified bodies scrutinize these justifications closely. A vague or templated rationale is one of the fastest ways to draw a major finding during a notified body audit.
Frequently Asked Questions
What is post-market clinical follow-up (PMCF)?
PMCF is a mandatory, continuous process under EU MDR Annex XIV, Part B, where manufacturers proactively collect and evaluate clinical data from CE-marked devices after market release. It keeps the Clinical Evaluation Report current and confirms safety and performance throughout the device lifecycle.
What is covered under post-market clinical follow-up?
PMCF covers all activities aimed at confirming device safety and performance, updating the benefit-risk assessment, and detecting emerging risks or off-label use patterns. This includes general methods (literature reviews, user feedback) and specific methods (prospective studies, registry evaluations).
What PMCF methods are used for post-market data collection (for example, surveys)?
Methods include literature reviews, adverse event database screening, healthcare professional and patient surveys, device registry analysis, and prospective post-market clinical studies. The choice and rigor of methods must be proportionate to the device's risk class and identified clinical data gaps.
What is the difference between PMCF and PMS?
PMS (Post-Market Surveillance) is the broader system covering all post-market activities including vigilance, complaint handling, and trend reporting. PMCF is a clinical-data-focused subset of PMS specifically designed to update the CER. PMCF findings feed back into clinical evaluation, while broader PMS data informs corrective actions.
Does ISO 13485 require post-market surveillance, and which ISO standard covers it?
ISO 13485 (Section 8.2.1) requires manufacturers to establish documented procedures for post-market surveillance as part of their QMS. It sets the system-level requirement; specific PMCF content and methods are governed by EU MDR Annex XIV and MDCG guidance, not a separate ISO standard.
What is the performance evaluation plan for the IVDR?
Under IVDR, the equivalent of PMCF is Post-Market Performance Follow-Up (PMPF), governed by IVDR Annex XIII, Part B. PMPF requires a PMPF Plan outlining methods to confirm the safety, performance, and scientific validity of IVD devices throughout their lifecycle, feeding results into the Performance Evaluation Report (PER).


