
Introduction
Receiving an FDA warning letter triggers a high-stakes regulatory countdown. Companies have just 15 working days to submit a written response that satisfies agency expectations. The quality of that response determines whether the situation resolves with a close-out letter or escalates to product seizures, injunctions, or criminal referrals.
FDA issued 303 warning letters to drug and biologics firms in FY2025 alone — a 59% increase from FY2024's 190 letters. Medical device, pharmaceutical, and IVD manufacturers face tighter scrutiny and faster enforcement action in 2026, with the FDA cutting warning letter issuance time by approximately one-third.

Every warning letter is also publicly posted on the FDA's searchable database — reputational exposure that compounds the operational pressure.
Choosing the right FDA warning letter response consultant matters. Look for a team with:
- CAPA expertise and structured root cause analysis methodology
- Direct FDA communication experience — not just internal compliance work
- A track record of close-out letters, not just submitted responses
- Industry-specific knowledge in your product category (device, pharma, IVD)
TL;DR
- An FDA warning letter requires a written response within 15 working days; inadequate responses can escalate to seizures, injunctions, or penalties
- The right consultant combines CAPA expertise, root cause analysis, and sector-specific FDA communication experience
- Prioritize consultants with proven audit clearance rates, industry-specific FDA experience, and full-cycle response support
- Rankings factor in enforcement action track records, service depth, and verified client outcomes across medical device, pharma, and IVD sectors
- Elexes leads with 50+ years of collective experience and a 90% audit clearance rate across 250+ global projects
What Is an FDA Warning Letter Response Consultant?
An FDA warning letter response consultant helps regulated companies — medical devices, pharmaceuticals, food, and diagnostics — analyze cited violations, conduct root cause analysis, and draft CAPA plans. They also manage the written response back to the FDA, ensuring corrective actions are communicated clearly and completely.
When you need one:
- Your internal team lacks FDA enforcement experience
- Violations span multiple sites or product lines
- A previous 483 response was deemed inadequate and escalated to a warning letter
- The 15-day response window leaves no margin for error
Skilled consultants also help prevent warning letters by ensuring thorough 483 responses — making this a proactive compliance investment, not just a crisis response. According to the FDA Regulatory Procedures Manual, inadequate 483 responses are a documented escalation pathway to formal enforcement.
Top 10 FDA Warning Letter Response Consultants in 2026
These consultants were selected based on FDA regulatory expertise, sector-specific experience, responsiveness, CAPA and QMS capabilities, and a track record of helping clients receive FDA close-out letters.
Elexes
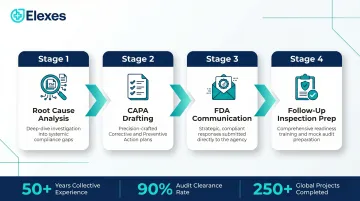
Elexes is a medical device regulatory consulting firm with 50+ years of collective experience, 250+ successful projects, a 90% audit clearance rate, and 100+ global clients across FDA, EU MDR, Health Canada, and other regulatory frameworks.
Key differentiators:
- End-to-end FDA warning letter response support: root cause analysis → CAPA drafting → FDA communication → follow-up inspection prep
- Cross-functional team covering regulatory, clinical, and QMS disciplines
- Flexible engagement models: full-time, part-time, or project-based
- Complete data confidentiality commitment
| Attribute | Details |
|---|---|
| Industry Focus | Medical Devices, IVDs, SaMD, Combination Products, Wearables, Digital Health |
| Key Services | FDA Warning Letter Response, 483 Response, CAPA Development, QMS Support, Regulatory Due Diligence |
| Ideal For | Startups to enterprise medical device companies seeking end-to-end compliance support globally |
mdi Consultants
mdi Consultants is a healthcare industry consulting firm with expertise in FDA compliance for medical devices, pharmaceuticals, and food companies, featuring a dedicated crisis intervention service line for warning letter response.
Key differentiators:
- Long-standing FDA regulatory experience
- Dedicated crisis intervention service line for companies receiving warning letters or facing imminent audits
- Legal counsel coordination for warning letter situations
| Attribute | Details |
|---|---|
| Industry Focus | Medical Devices, Pharmaceuticals, Food, IVDs, Biotechnology |
| Key Services | Warning Letter Response, 483 Consulting, FDA Compliance, Crisis Intervention, ISO/MDSAP |
| Ideal For | Companies needing combined regulatory and legal support for FDA enforcement actions |
J&J Compliance Consulting Group
J&J Compliance Consulting Group is an FDA regulatory compliance firm serving medical devices, pharmaceuticals, dietary supplements, food and beverage, cosmetics, and tobacco industries, with a dedicated FDA warning letter response practice.
Key differentiators:
- Multi-industry FDA coverage spanning five regulated sectors
- Structured warning letter guidance process with SMART criteria CAPA plans
- QMS improvement tied directly to CAPA development
| Attribute | Details |
|---|---|
| Industry Focus | Medical Devices, Pharma, Dietary Supplements, Cosmetics, Food and Beverage |
| Key Services | FDA Warning Letter Help, CAPA Planning, QMS Training, Regulatory Compliance Services |
| Ideal For | Multi-product or multi-industry companies facing cross-sector FDA enforcement |
Manex Consulting
Manex Consulting is a California-based manufacturing excellence consulting firm with experience helping organizations respond to FDA Form 483s and warning letters, particularly in food, pharma, and device manufacturing.
Key differentiators:
- Strong regional presence in California
- Integrated quality and operational consulting (not just regulatory)
- Practical CAP development with 30+ years of manufacturing operational experience
| Attribute | Details |
|---|---|
| Industry Focus | Food & Beverage, Medical Devices, Pharmaceuticals, General Manufacturing |
| Key Services | FDA 483 and Warning Letter Response, Corrective Action Plans, cGMP Compliance, ISO 9001 |
| Ideal For | California-based or U.S. manufacturers needing combined operational and regulatory corrective action support |
The FDA Group
The FDA Group is a regulatory and compliance staffing and consulting firm specializing in FDA-regulated industries, featuring a dedicated warning letter recovery practice for life sciences companies and a network of 325+ former FDA employees across 3,750+ resources.
Key differentiators:
- Deep bench of former FDA officials and industry veterans
- Rapid-response staffing model deploying embedded experts for warning letter situations
- Documented warning letter recovery methodology with former FDA investigators who have written hundreds of 483 observations
| Attribute | Details |
|---|---|
| Industry Focus | Pharmaceuticals, Biotechnology, Medical Devices, Life Sciences |
| Key Services | Warning Letter Recovery, CAPA Consulting, Regulatory Staffing, FDA Audit Preparation |
| Ideal For | Life sciences companies needing rapid resource deployment after receiving an FDA enforcement action |
Greenlight Guru
Greenlight Guru is a medical device quality management software and consulting platform that supports FDA 483 and warning letter response through its eQMS tools and regulatory expert network. Important distinction: Greenlight Guru is a software platform with advisory services, not a traditional consulting firm.
Key differentiators:
- Purpose-built medical device QMS software with CAPA workflows
- Structured quality event tracking and document management
- Access to regulatory consultants through Guru Advisory Services (Guru Assist, Guru Guidance, Professional Services)
| Attribute | Details |
|---|---|
| Industry Focus | Medical Devices, Digital Health, SaMD |
| Key Services | eQMS, CAPA Management, FDA 483/Warning Letter Guidance, Document Control |
| Ideal For | Medical device companies wanting a software-driven approach to CAPA and warning letter response |
Regulatory Compliance Associates (RCA)
Regulatory Compliance Associates is a global regulatory consulting firm (a Nelson Labs / Sotera Health company) with extensive FDA experience across pharmaceuticals, biologics, and medical devices, offering warning letter response as part of a broader compliance remediation practice. RCA serves 40 of the top 50 medical device companies and 8 of the top 10 pharmaceutical companies.
Key differentiators:
- Global regulatory reach with a network of experts in 50+ countries
- Former FDA reviewer expertise on staff
- Comprehensive remediation services extending beyond immediate warning letter response
| Attribute | Details |
|---|---|
| Industry Focus | Pharmaceuticals, Biologics, Medical Devices, OTC Products |
| Key Services | Warning Letter Response, Regulatory Remediation, FDA Inspection Readiness, Quality Systems |
| Ideal For | Large pharma and biologics companies managing complex or multi-site warning letter remediation |
PAREXEL Consulting
PAREXEL is a global CRO and regulatory consulting firm with a dedicated regulatory affairs practice that supports warning letter response and consent decree remediation for large pharmaceutical and device manufacturers. The team includes 80+ ex-agency regulators and access to nearly 2,500 sites globally.
Key differentiators:
- Global scale with experience across 40+ years
- Integrated clinical and regulatory expertise
- Ability to handle large-scale remediation programs involving multiple regulatory bodies simultaneously
| Attribute | Details |
|---|---|
| Industry Focus | Pharmaceuticals, Biotechnology, Medical Devices, Diagnostics |
| Key Services | Warning Letter Response, Consent Decree Remediation, Regulatory Strategy, CAPA Programs |
| Ideal For | Enterprise-level companies with complex, multi-jurisdictional FDA enforcement situations |
Halloran Consulting Group
Halloran Consulting Group is a life sciences consulting firm with specialized expertise in FDA regulatory strategy, quality systems, and compliance remediation, including GxP Quality Assessment services.
Key differentiators:
- Focused life sciences specialization across biotech, pharma, devices, and digital health
- Strong FDA strategic advisory capabilities
- QMS-linked corrective action support with tailored solutions
| Attribute | Details |
|---|---|
| Industry Focus | Pharmaceuticals, Biotechnology, Medical Devices |
| Key Services | FDA Warning Letter Response, Quality Remediation, Regulatory Strategy, 483 Response |
| Ideal For | Biotech and pharma companies needing strategic FDA advisory alongside warning letter response |
Biologics Consulting Group
Biologics Consulting Group is an FDA regulatory consulting firm serving pharmaceutical, biological, and medical device companies, with services spanning warning letter response, CAPA, and pre-submission strategy. Founded in 1993 by former FDA officers, the firm operates as a fully employee-owned (ESOP) company staffed by industry experts and former FDA officials.
Key differentiators:
- Deep biologics and combination product expertise
- Former FDA regulatory reviewers on staff providing insider perspective
- Structured approach to warning letter remediation
| Attribute | Details |
|---|---|
| Industry Focus | Biologics, Pharmaceuticals, Combination Products, Medical Devices |
| Key Services | Warning Letter Response, CAPA Planning, Regulatory Submissions, Pre-Inspection Audit Support |
| Ideal For | Biologics and combination product manufacturers navigating complex FDA enforcement actions |
How We Chose the Best FDA Warning Letter Response Consultants
Consultants were evaluated on sector-specific FDA experience — not generic compliance knowledge — and the depth of their warning letter and Form 483 response capabilities. That includes CAPA development, root cause analysis, FDA communication, and follow-up inspection preparation. Outcome evidence mattered too: audit clearance rates, close-out letter attainment, and documented resolution timelines.
Common mistakes companies make:
- Selecting based on brand name alone without verifying FDA warning letter-specific experience
- Choosing firms that outsource actual response writing to subcontractors
- Engaging consultants who lack experience in the specific regulated industry (e.g., choosing a food safety consultant for a medical device warning letter)
Additional evaluation factors:
- Responsiveness and ability to mobilize within the 15-working-day window
- Transparent engagement models: fixed fee vs. hourly, full-time vs. part-time
- Ongoing compliance support beyond the immediate response to help prevent future enforcement actions

That last point connects directly to what the FDA actually requires. According to the FDA's guidance on close-out letters, the agency will not issue a close-out letter based on "representations" alone — corrections must be implemented and verified, typically through a follow-up inspection. Consultants who understand this end-to-end verification pathway are far more likely to get you to a formal close-out, not just a submitted response.
Conclusion
Receiving an FDA warning letter is not the end, but your response quality determines whether the situation resolves quickly or escalates to injunctions, seizures, or criminal referrals. Choosing the right consultant is one of the most consequential decisions you'll make in the process.
When evaluating your options, prioritize fit over name recognition. Key criteria to weigh:
- Industry match: Medical device, pharma, and food violations require different regulatory expertise
- Violation complexity: Single procedural gaps differ from systemic QMS failures
- Remediation scope: Some firms handle response drafting only; others rebuild the underlying quality system
- Regulatory track record: Ask for documented outcomes, not just client counts
For medical device companies, IVD manufacturers, or digital health developers facing an FDA warning letter or 483 response, Elexes offers a team with 50+ years of collective experience, a 90% audit clearance rate, and flexible engagement models. Reach out to discuss your specific situation.
Frequently Asked Questions
How to find FDA warning letters?
FDA warning letters are publicly available on the FDA's official website under the "Warning Letters" section of the ICECI database. You can search by company name, product type, issue date, and industry — and reviewing letters in your sector is a useful compliance benchmarking exercise.
What is the difference between a 483 and a warning letter?
A Form 483 is a list of inspectional observations issued by an FDA investigator at the end of an inspection, giving the company an opportunity to respond voluntarily. A Warning Letter is a formal escalation issued by senior FDA leadership when violations are deemed significant or when the 483 response was inadequate. Warning letters carry greater legal weight, public visibility, and enforcement consequences.
How long do you have to respond to an FDA warning letter?
Companies typically have 15 working days to submit a written response to an FDA warning letter. While the FDA may occasionally grant extensions for complex cases, this should not be relied upon. The response must include a CAPA plan, root cause analysis, and evidence of corrective actions already underway.
What should an FDA warning letter response include?
A compliant response must address each cited violation with documented root cause analysis, a detailed CAPA plan with timelines, and supporting evidence such as revised SOPs, training records, or validation reports. It should also include a commitment from senior leadership and a schedule for reporting ongoing corrective progress to the FDA.
Can hiring an FDA warning letter response consultant help prevent future warning letters?
Yes. A good consultant not only helps craft the immediate response but also identifies systemic gaps in the QMS, cGMP practices, or documentation that triggered the violation. Addressing these root causes and strengthening ongoing compliance monitoring significantly reduces the risk of future 483 observations or warning letters.
What happens if you do not respond to an FDA warning letter?
Failure to respond — or submitting an inadequate response — can trigger formal FDA enforcement actions: product seizures, manufacturing injunctions, civil penalties, or mandatory recalls. In severe cases, company executives may face criminal charges.


