
Introduction
FDA inspections have tightened significantly across medical device, pharmaceutical, and biotech companies. Between July and December 2025 alone, the FDA issued 327 warning letters—a 73% increase over the same period in 2024. Drug quality inspections surged from 766 (FY2023) to 972 (FY2024), while device inspections grew from approximately 2,150 to 2,330. A single unresolved compliance gap can trigger a Form 483, Warning Letter, or costly product recall—making professional readiness support not just helpful but strategically critical.
What drives most inspection failures isn't science — it's operational gaps. The FDA's most frequently cited observations cluster around three recurring problem areas:
- CAPA deficiencies (21 CFR 820.100(a)) — incomplete corrective action documentation and root cause analysis
- Complaint handling failures (21 CFR 820.198(a)) — inadequate intake, investigation, and closure processes
- Process validation gaps — missing or insufficient evidence that manufacturing processes consistently produce conforming product
Specialist consultants address exactly these gaps, shifting companies from reactive scrambling to structured, audit-ready compliance programs.
This guide profiles the top 10 FDA inspection readiness consultants in 2026, selected based on demonstrated audit expertise, clearance track records, service depth, and global reach across medical device and life sciences sectors.
TL;DR
- FDA inspection readiness consulting closes compliance gaps and helps companies avoid 483 observations
- Top consultants deliver mock inspections, gap assessments, CAPA remediation, and inspector-behavior training — not just reports
- Elexes brings 50+ years of collective experience, a 90% audit clearance rate, and coverage across FDA, ISO 13485, and EU MDR
- Choose based on industry-specific expertise, proven outcomes, and engagement flexibility — not brand name alone
- Firms below range from specialist boutiques to global platforms, serving early-stage startups to multinational manufacturers
What Is FDA Inspection Readiness Consulting?
FDA inspection readiness consulting is the structured process of preparing a medical device or life sciences organization to withstand an unannounced or scheduled FDA audit. It encompasses documentation review, QMS gap analysis, mock inspections, personnel behavioral training, and CAPA remediation—covering the full spectrum of compliance risk.
The stakes are high. The medical device industry incurs approximately $9 billion annually from 483 observations, recalls, warning letters, consent decrees, and legal fees. A single recall costs nearly $10 million, excluding lost sales and reputational damage. With the FDA deploying AI-driven risk targeting through its internal system "Elsa"—which analyzes historical Form 483 observations, compliance anomalies, and adverse event data—companies can no longer rely on low inspection probability. Targeted, evidence-based inspections are intensifying.
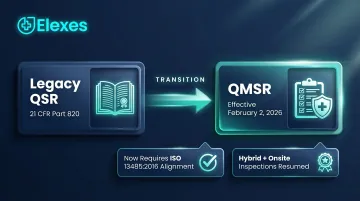
The regulatory environment has also shifted structurally. The Quality Management System Regulation (QMSR) became effective February 2, 2026, replacing the legacy QSR and incorporating ISO 13485:2016 by reference. Every medical device manufacturer must now demonstrate ISO 13485 alignment during inspections. With FDA inspections resuming at full volume—including hybrid and onsite formats—companies increasingly rely on third-party inspection readiness consultants to meet these dual compliance demands.
Top 10 FDA Inspection Readiness Consultants in 2026
The following 10 consultants were selected based on demonstrated FDA audit expertise, QMS and regulatory certifications, client outcomes, industry specialization, and their capacity to deliver both pre-inspection readiness and post-inspection remediation.
Elexes Medical Consulting
Elexes is a globally recognized medical device regulatory consulting firm with 50+ years of collective experience, having completed 250+ successful projects across 200+ product types for 100+ clients in the US, UK, Europe, Canada, Australia, and Asia—with a 90% audit clearance rate. Core FDA inspection readiness services include QMS gap assessments, mock FDA inspections, CAPA support, ISO 13485 compliance, and regulatory due diligence.
Elexes differentiates through a cross-functional outsourcing model that combines regulatory, quality, and clinical tasks under flexible full-time, part-time, or project-based engagements. Its consultant team covers FDA, EU MDR, Health Canada, EMA, and MHRA frameworks. Clients such as AliveCor, Outset Medical, DJOGlobal, and Radformation reflect the firm's reach across device categories from wearables to SaMD and implants.
| Key Services | Mock FDA inspections, QMS gap assessments, CAPA remediation, regulatory due diligence, ISO 13485/MDSAP compliance, clinical trial documentation, post-market surveillance |
|---|---|
| Industry Focus | Medical devices, IVD/LDT, SaMD, wearables, implants, combination devices, diagnostics, digital health |
| Notable Credentials | ISO 13485, ISO 9001, MDSAP, GCP, GLP, HIPAA; 90% audit clearance rate; 50+ years collective experience |

Regulatory Compliance Associates (RCA)
RCA, a Nelson Labs/Sotera Health company, is a specialized life sciences consulting firm with deep expertise in FDA compliance, audits, QMS remediation, and inspection readiness for medical device and pharmaceutical companies. It supports clients across the full product lifecycle from pre-approval to post-market.
RCA differentiates itself through a proprietary inspection readiness methodology that includes front-room/back-room management, behavioral training, and structured personnel roles (Inspection Coordinators, System Owners, Escorts, Scribes). The firm serves 5,000+ customers in over 50 countries and is ISO 9001 certified.
RCA claims to support 40 of the top 50 medical device companies and 8 of the top 10 pharmaceutical companies, though these figures are self-reported on the company's website.
| Key Services | Compliance training and inspection readiness, audits and assessments, QMS remediation, regulatory affairs, data integrity consulting |
|---|---|
| Industry Focus | Medical devices, pharmaceuticals, biologics, combination products, 503 compounders |
| Notable Credentials | ISO 9001 certified; part of Sotera Health group; claims to serve 40 of the top 50 medical device companies |
BioBoston Consulting
BioBoston Consulting is a Boston-based life sciences consultancy that claims to have received the "Global Excellence Award – Best Life Science Business Consultancy" in 2024 and 2025. It focuses on FDA inspection readiness, GxP compliance assessments, QMS strengthening, and regulatory submission readiness for biotech, pharma, and medtech companies. The firm reports a team of 650+ senior professionals, including former FDA investigators.
BioBoston emphasizes a risk-based compliance strategy approach and has built a reputation for startup-focused readiness programs. However, the awarding body for the "Global Excellence Award" could not be independently verified, and the claim appears only on BioBoston's own content.
| Key Services | FDA inspection readiness, mock inspections, GxP compliance assessments, internal audits, QMS enhancement, data integrity consulting |
|---|---|
| Industry Focus | Biotech, pharmaceutical, medical devices, diagnostics |
| Notable Credentials | Claims "Global Excellence Award" 2024 and 2025 (unverified); 650+ senior professionals |
IQVIA
IQVIA is a global data and consulting company serving pharmaceutical, biotech, and medical device industries, with approximately 93,000 employees in over 100 countries and 2023 revenue of $14.98 billion. Within FDA inspection readiness, IQVIA focuses on clinical data integrity, real-world evidence alignment, regulatory data analytics, and post-market safety monitoring compliance.
IQVIA's SmartSolve platform—built on Microsoft Azure—offers AI-powered quality management and regulatory information management. The suite includes eQMS, regulatory information management (RIM), and compliance modules. SmartSolve won a 2023 MedTech Breakthrough Award and a Frost & Sullivan Award, and IQVIA claims zero regulatory inspection findings in its operations.
| Key Services | Clinical trial optimization, regulatory data analytics, post-market safety monitoring, compliance program development, AI-powered eQMS |
|---|---|
| Industry Focus | Pharmaceutical, biotech, medical devices |
| Notable Credentials | 93,000 employees; $14.98B revenue (2023); SmartSolve MedTech Breakthrough Award; zero inspection findings claimed |
Emergo by UL
Emergo by UL is a dedicated medical device regulatory consulting firm offering specialized FDA inspection readiness, 510(k), De Novo, and PMA submission support, quality system consulting, and global market access services. It operates from 20+ offices on six continents, serving medical device and IVD companies in more than 20 countries since 1997.
Emergo provides a digital platform called RAMS (Regulatory Affairs Management Suite) to automate RA/QA activities, plus OPUS for human factors engineering. The firm supports ISO 13485, FDA QSR, EU MDR/IVDR, MDSAP, UKCA, Health Canada, and ANVISA frameworks, making it a strong choice for device-specific compliance needs.
| Key Services | FDA inspection readiness, 510(k)/PMA/De Novo support, QMS consulting, ISO 13485 implementation, global regulatory submissions |
|---|---|
| Industry Focus | Medical devices, IVDs, SaMD, combination products |
| Notable Credentials | Part of UL group; 20+ offices on six continents; RAMS and OPUS platforms; ISO certificate listed |
Greenlight Guru
Greenlight Guru is a quality management software and consulting platform purpose-built for medical device companies. It supports FDA inspection readiness by helping teams build and maintain compliant QMS documentation, design history files, and audit-ready records in real time. Unlike traditional consultants, Greenlight Guru leads with a cloud-based eQMS platform.
The platform is built around FDA and ISO standards with audit-tested workflows covering:
- Quality management and CAPA tracking
- Product development (requirements, risk per ISO 14971, software release)
- Clinical evidence (EDC, ePRO/eCOA, PMCF)
- Supplier qualification and parts management
It holds ISO 9001 and ISO 27001 certifications and features Halo, an AI and ML recommendation engine for quality and change management. Greenlight Guru received a $120 million investment from JMI Equity in 2021 and has strategic alliances with Nelson Labs, Emergo by UL, and Reed Tech.
| Key Services | QMS software implementation, design history file management, CAPA tracking, audit trail documentation, inspection readiness consulting |
|---|---|
| Industry Focus | Medical devices (all classes), SaMD, IVD |
| Notable Credentials | ISO 9001 and ISO 27001 certified; $120M JMI Equity investment; Halo AI engine; software-first platform |
Deloitte Life Sciences
Deloitte's Life Sciences practice provides FDA compliance transformation, risk management, GxP process improvement, and quality system modernization for pharmaceutical, biotech, and medical device companies. It serves 90% of Fortune 500 life sciences companies, nearly 95% of medical device manufacturers, and the top 12 largest pharmaceutical companies.
Deloitte's service lines span compliance strategy, design, and operations; digital quality and compliance (SaMD management, QMS assessment, CSV managed services); investigations and settlement support (CIA readiness, IRO services); and HCP interactions and payments.
Proprietary technology includes InteractRx (FMV physician compensation) and Spark (interactive digital compliance platform), with AI integration for HCP transparency.
| Key Services | FDA regulatory compliance frameworks, GxP improvements, risk management systems, quality and compliance transformation, digital quality and compliance |
|---|---|
| Industry Focus | Pharmaceutical, biotech, medical devices, diagnostics |
| Notable Credentials | Serves 90% of Fortune 500 life sciences firms, 95% of medical device manufacturers; InteractRx and Spark platforms |
Accenture Life Sciences
Accenture's Life Sciences group focuses on digital transformation for regulatory compliance—including AI-assisted documentation workflows, digital QMS platforms, cloud-based regulatory submission management, and data analytics for compliance monitoring. The practice has more than 22,000 dedicated life sciences professionals, with 250+ skilled regulatory professionals, experience in 90+ countries, and 200+ clients.
Accenture is a Veeva RIM Partner with 5+ enterprise-scale RIM implementations for Tier-1 clients. Its proprietary tools include iACTIVATE (AI-based artwork proofreading tool reporting 40% efficiency gain) and generative AI for LLM-based submission authoring. Accenture claims zero regulatory inspection findings in its operations.
| Key Services | Digital QMS implementation, AI-supported documentation, cloud-based regulatory workflows, compliance analytics, Veeva RIM implementations |
|---|---|
| Industry Focus | Pharmaceutical, biotech, medical devices, digital health |
| Notable Credentials | 22,000 life sciences professionals; 250+ regulatory experts; Veeva RIM Partner; zero inspection findings claimed |
KPMG Life Sciences
KPMG's Life Sciences advisory practice supports FDA inspection readiness through compliance governance frameworks, internal audit preparation, regulatory risk assessments, and corrective action programs—particularly valuable for companies managing complex multi-site or multi-jurisdiction compliance programs.
KPMG provides services across the full regulatory compliance lifecycle: compliance program development and governance, risk mitigation controls, monitoring and benchmarking, investigation and litigation support, and regulatory affairs services including process and technology implementation. KPMG was the first Big Four firm to organize along industry lines, including dedicated Healthcare and Life Sciences practices. Practice-level client volume figures are not publicly available.
| Key Services | Compliance program development, internal audit preparation, regulatory risk frameworks, corrective action support, governance structure design |
|---|---|
| Industry Focus | Pharmaceutical, biotech, medical devices, life sciences manufacturing |
| Notable Credentials | First Big Four firm organized by industry; scale data not publicly disclosed |
PwC Life Sciences
PwC's Life Sciences division advises pharmaceutical and medical device organizations on regulatory risk management, compliance governance, global regulatory strategy, and market expansion—making it particularly valuable for multinational companies managing simultaneous FDA and international regulatory obligations.
PwC's Pharma and Life Sciences practice covers six functional areas: research and development, commercial operations, supply chain and manufacturing, medical affairs, regulatory/compliance/quality, and corporate and financial strategy. The firm emphasizes AI, emerging tech, and "digital-first workforce" approaches. PwC operates through a global network with specialists advising at global, national, and local levels. Specific client counts or practice size data were not publicly disclosed.
| Key Services | Regulatory risk assessments, internal audit frameworks, compliance governance structures, global regulatory strategy, market expansion support |
|---|---|
| Industry Focus | Pharmaceutical, biotech, medical devices, diagnostics |
| Notable Credentials | Global network; six functional areas; AI and digital-first focus; scale data not publicly disclosed |
How We Chose the Best FDA Inspection Readiness Consultants
These firms were assessed across six criteria:
- FDA audit expertise: mock inspection capability, CAPA remediation, QMS gap analysis
- Industry-specific track records: medical devices vs. broad life sciences
- Relevant certifications: ISO 13485, MDSAP, GLP, GCP
- Client outcomes: audit clearance rates and 483 remediation success
- Geographic reach: domestic and international coverage
- Engagement flexibility: project-based, retainer, and embedded models

These criteria exist because the most common selection mistakes come down to misaligned expectations—not lack of options.
Prioritizing brand name over specialized regulatory depth is a recurring misstep. Choosing a Big Four firm for its global presence when hands-on mock inspection capability is what's actually needed often leads to misaligned support. Conversely, selecting a boutique firm without enterprise-scale technology when managing multi-site, multi-jurisdiction compliance can create bottlenecks.
Companies that engage specialized FDA inspection readiness partners earlier in their product lifecycle report fewer observations, faster remediation timelines, and stronger long-term compliance cultures.
The financial stakes make early investment clear. The medical device industry incurs approximately $9 billion annually in costs from observations, recalls, and enforcement actions. Proactive readiness is a fraction of the cost of reactive remediation—particularly as DOJ False Claims Act recoveries totaled $6.8 billion in a recent fiscal year, with medical device companies as primary targets.
Conclusion
FDA inspection readiness is a continuous compliance discipline, not a one-time pre-inspection exercise. It requires systematic QMS maintenance, documentation integrity, CAPA effectiveness, and cross-functional alignment throughout the product lifecycle. With the QMSR now in effect and FDA inspections driven by AI-based risk targeting, treating readiness as an ongoing strategic priority is no longer optional.
When selecting a consultant, look beyond reputation. Evaluate based on:
- Sector expertise specific to medical devices, IVDs, SaMD, or your product category
- Audit clearance track record — measurable outcomes, not just references
- Engagement flexibility — project-based, retainer, or embedded support options
- Internal integration — ability to work alongside your team without disrupting operations
Specialist consultants are best suited for tactical readiness ahead of imminent inspections. Enterprise-scale firms are better positioned for systemic quality transformation across global networks.
For a specialized partner with a 90% audit clearance rate and end-to-end regulatory support — from QMS implementation to inspection response — contact Elexes to discuss your compliance needs.
Frequently Asked Questions
How much do FDA inspection readiness consultants charge?
Fees vary based on scope, firm size, and engagement model. Project-based work for small companies typically ranges from $15,000–$50,000; enterprise retainer programs can exceed $250,000 annually. Hourly rates run $200–$400 for specialist consultants and $300–$600+ at Big Four firms depending on seniority.
How long do FDA audits take?
Audit duration depends on inspection type and facility size. Routine device inspections typically last 1–5 business days, while complex pre-approval or for-cause inspections can run longer. Hybrid and records-request formats may span several weeks.
What are the 4 types of FDA inspections?
The four types are:
- Surveillance — routine monitoring of manufacturing processes and CGMP compliance
- Application-Based — pre-approval inspections assessing compliance and data integrity before market clearance
- For-Cause — triggered by suspected quality problems or complaints
- Compliance Follow-Up — verifying corrective actions after prior violations
Each type carries distinct risk levels and preparation requirements.
What is the difference between FDA 483 and EIR?
A Form 483 lists "significant objectionable conditions" observed during an inspection and is issued directly to firm management at its close. An Establishment Inspection Report (EIR) is the FDA's internal document prepared afterward, summarizing scope, evidence, and conclusions — it is not provided to the company.
Who performs FDA inspections?
FDA inspections are conducted by Consumer Safety Officers (investigators) employed by the FDA's Office of Regulatory Affairs (ORA). For complex technologies, investigators may be accompanied by specialists from CDRH (devices) or CDER (drugs). Device-specific inspections reference 21 CFR Part 820 and the new QMSR, which incorporates ISO 13485:2016 by reference.


